1. Pateder DB, Kebaish KM, Cascio BM, Neubaeur P, Matusz DM, Kostuik JP. Posterior only versus combined anterior and posterior approaches to lumbar scoliosis in adults: a radiographic analysis. Spine (Phila Pa 1976) 2007;32:1551ŌĆō4.


2. Dorward IG, Lenke LG, Bridwell KH, et al. Transforaminal versus anterior lumbar interbody fusion in long deformity constructs: a matched cohort analysis. Spine (Phila Pa 1976) 2013;38:E755ŌĆō62.


3. Bergin PF, OŌĆÖBrien JR, Matteini LE, Yu WD, Kebaish KM. The use of spinal osteotomy in the treatment of spinal deformity. Orthopedics 2010;33:586ŌĆō94.


5. De Kunder SL, van Kuijk SM, Rijkers K, et al. Transforaminal lumbar interbody fusion (TLIF) versus posterior lumbar interbody fusion (PLIF) in lumbar spondylolisthesis: a systematic review and meta-analysis. Spine J 2017;17:1712ŌĆō21.


6. Ozgur BM, Aryan HE, Pimenta L, Taylor WR. Extreme lateral interbody fusion (XLIF): a novel surgical technique for anterior lumbar interbody fusion. Spine J 2006;6:435ŌĆō43.


7. Phillips FM, Isaacs RE, Rodgers WB, et al. Adult degenerative scoliosis treated with XLIF: clinical and radiographical results of a prospective multicenter study with 24-month follow-up. Spine (Phila Pa 1976) 2013;38:1853ŌĆō61.


10. Cheung KM, Luk KD. Prediction of correction of scoliosis with use of the fulcrum bending radiograph. J Bone Joint Surg Am 1997;79:1144ŌĆō50.


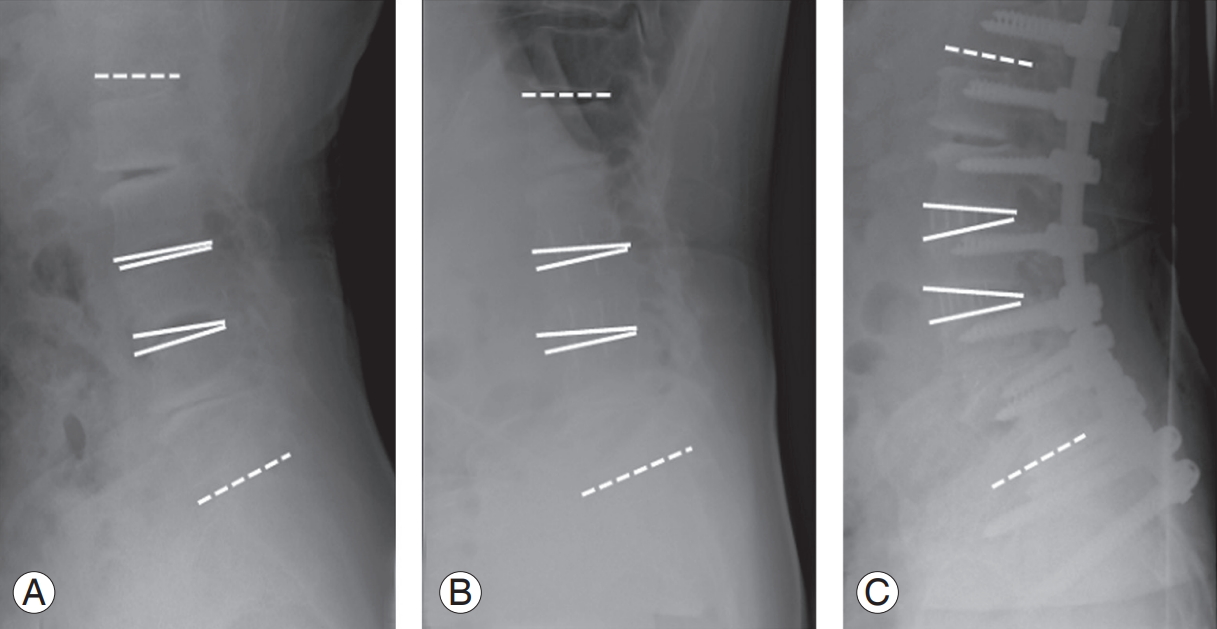
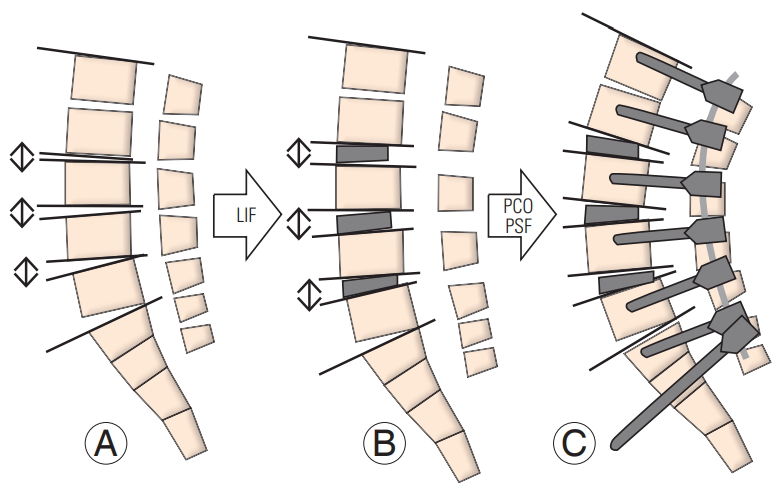
11. Matsumura A, Namikawa T, Kato M, et al. Posterior corrective surgery with a multilevel transforaminal lumbar interbody fusion and a rod rotation maneuver for patients with degenerative lumbar kyphoscoliosis. J Neurosurg Spine 2017;26:150ŌĆō7.


13. Oliveira L, Marchi L, Coutinho E, Pimenta L. A radiographic assessment of the ability of the extreme lateral interbody fusion procedure to indirectly decompress the neural elements. Spine (Phila Pa 1976) 2010;35(26 Suppl): S331ŌĆō7.


14. Kepler CK, Sharma AK, Huang RC, et al. Indirect foraminal decompression after lateral transpsoas interbody fusion. J Neurosurg Spine 2012;16:329ŌĆō33.


16. Sharma AK, Kepler CK, Girardi FP, Cammisa FP, Huang RC, Sama AA. Lateral lumbar interbody fusion: clinical and radiographic outcomes at 1 year: a preliminary report. J Spinal Disord Tech 2011;24:242ŌĆō50.


17. Mundis GM, Akbarnia BA, Phillips FM. Adult deformity correction through minimally invasive lateral approach techniques. Spine (Phila Pa 1976) 2010;35(26 Suppl): S312ŌĆō21.


18. Isaacs RE, Hyde J, Goodrich JA, Rodgers WB, Phillips FM. A prospective, nonrandomized, multicenter evaluation of extreme lateral interbody fusion for the treatment of adult degenerative scoliosis: perioperative outcomes and complications. Spine (Phila Pa 1976) 2010;35(26 Suppl): S322ŌĆō30.


19. Strom RG, Bae J, Mizutani J, Valone F 3rd, Ames CP, Deviren V. Lateral interbody fusion combined with open posterior surgery for adult spinal deformity. J Neurosurg Spine 2016;25:697ŌĆō705.


20. Park HY, Ha KY, Kim YH, et al. Minimally invasive lateral lumbar interbody fusion for adult spinal deformity: clinical and radiological efficacy with minimum two years follow-up. Spine (Phila Pa 1976) 2018;43:E813ŌĆō21.


21. Theologis AA, Mundis GM Jr, Nguyen S, et al. Utility of multilevel lateral interbody fusion of the thoracolumbar coronal curve apex in adult deformity surgery in combination with open posterior instrumentation and L5-S1 interbody fusion: a case-matched evaluation of 32 patients. J Neurosurg Spine 2017;26:208ŌĆō19.


23. Grubb SA, Lipscomb HJ, Coonrad RW. Degenerative adult onset scoliosis. Spine (Phila Pa 1976) 1988;13:241ŌĆō5.


24. Simmons ED. Surgical treatment of patients with lumbar spinal stenosis with associated scoliosis. Clin Orthop Relat Res 2001;(384): 45ŌĆō53.

25. Ponte A, Vero B, Sicicardi G,Surgical treatment of ScheuermannŌĆÖs kyphosis. Winter R, editors. Progress in spinal pathology: kyphosis. Bologna: Aulo Gaggi; 1984. p.75ŌĆō81.
26. Smith-Petersen MN, Larson CB, Aufranc OE. Osteotomy of the spine for correction of flexion deformity in rheumatoid arthritis. Clin Orthop Relat Res 1969;66:6ŌĆō9.


28. Yamato Y, Sato Y, Togawa D, et al. Differences in the geometrical spinal shape in the sagittal plane according to age and magnitude of pelvic incidence in healthy elderly individuals. J Orthop Sci 2019;S0949-2658(19): 30207ŌĆō6.