|
 |
- Search
| Asian Spine J > Volume 17(2); 2023 > Article |
|
Abstract
Purpose
Overview of Literature
Methods
Results
Notes
Acknowledgments
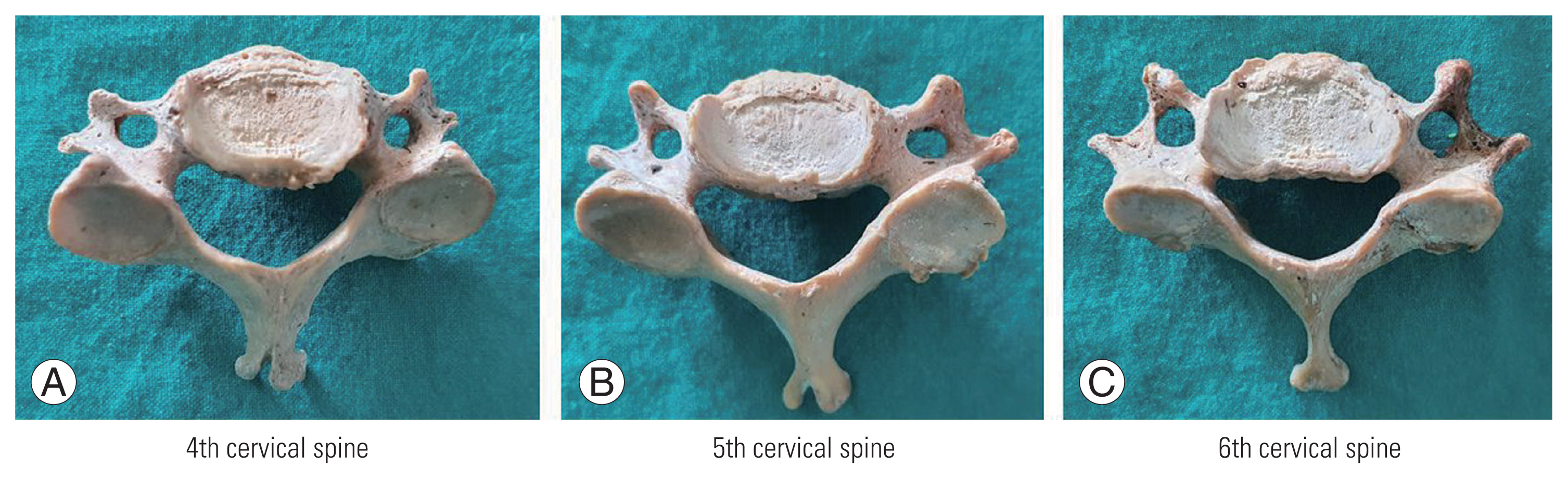
Fig. 2
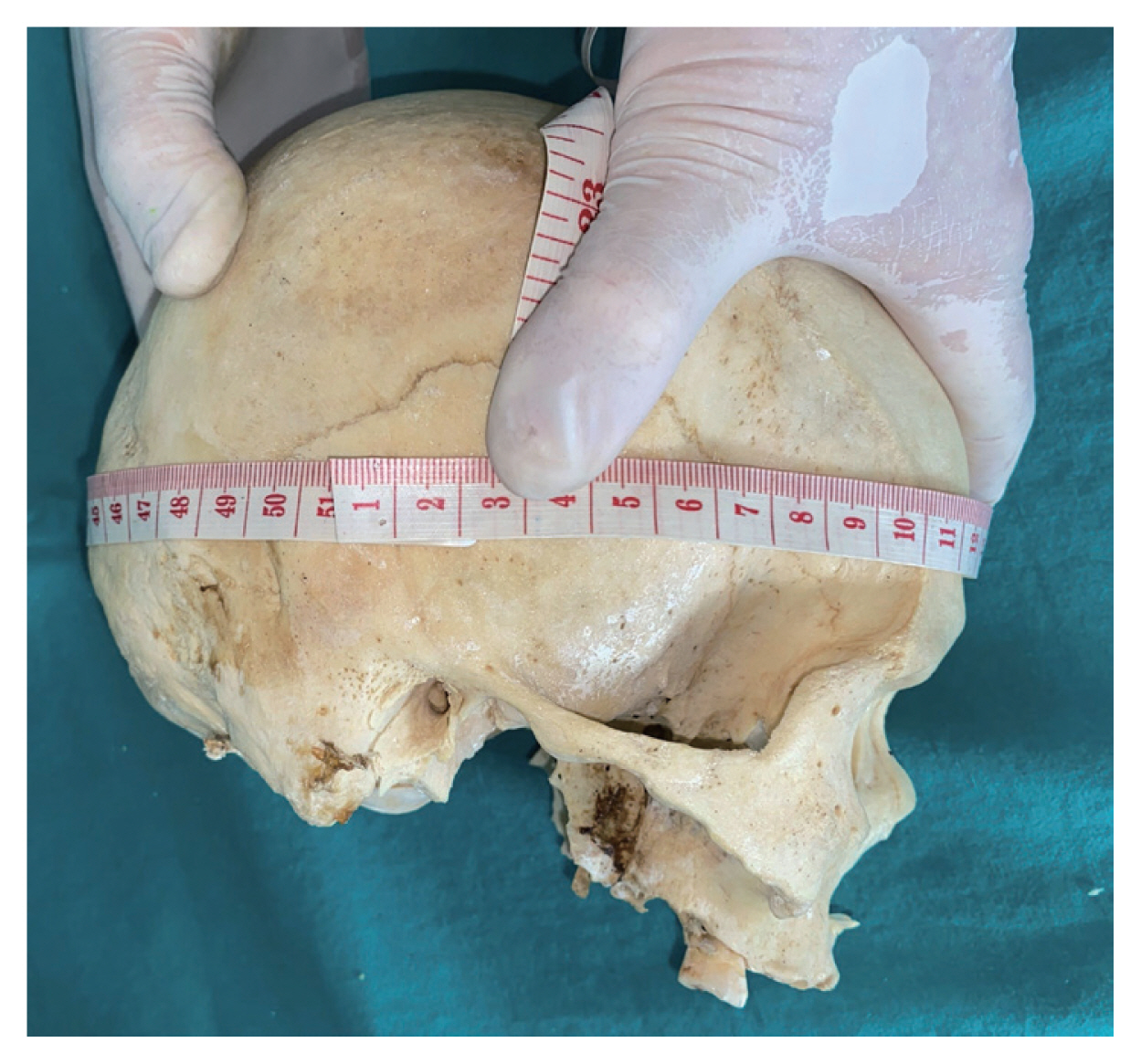
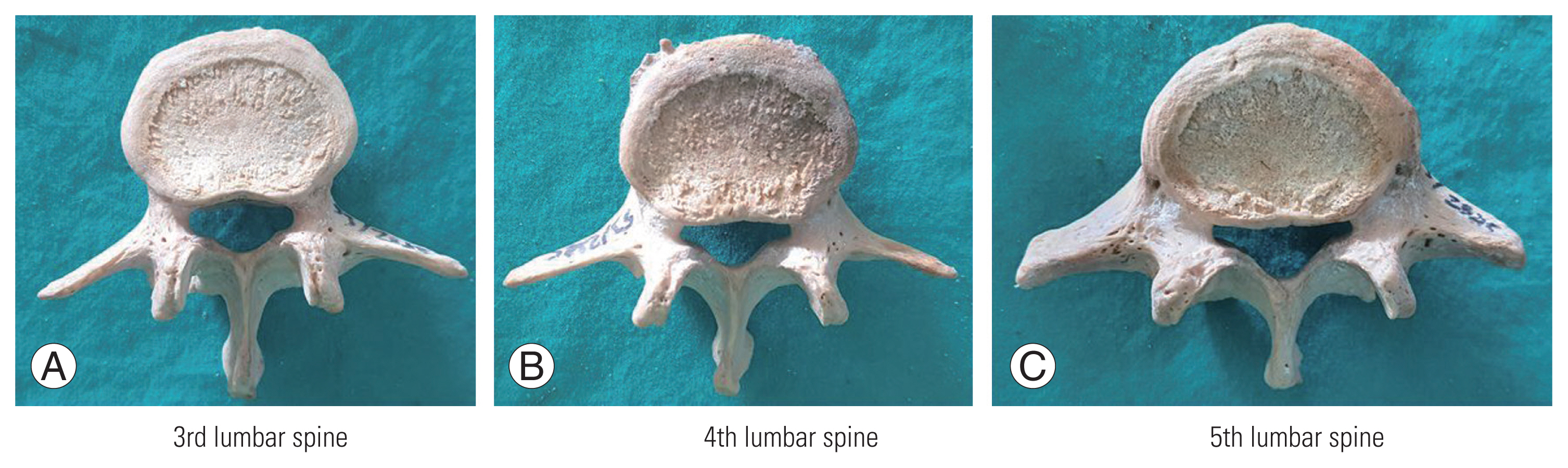
Fig. 3

Table 1
Table 2
| Variable | DSS (−) group | DCSS (+) group | DLSS (+) group | Tandem DSS (+) group | Significance (p-value) |
|---|---|---|---|---|---|
| No. of cases | 218 | 47 | 16 | 12 | |
| Age (yr) | 67.0±12.5 | 70.1±11.1 | 64.3±11.7 | 65.1±9.9 | NS (p=0.21221) |
| Skull circumference (cm) | 50.7±1.4a),b) | 49.1±1.1a) | 50.2±1.5 | 48.6±1.6b) | p<0.001 (p<0.00001) |
| AP diameter of foramen magnum (mm) | 35.4±2.4 | 34.5±2.2 | 34.6±1.3 | 34.0±1.4 | NS |
| Transverse diameter of foramen magnum (mm) | 30.4±2.4c),d) | 29.4±1.7c) | 29.6±1.7 | 28.2±2.2d) | p<0.01 (p=0.00114) |
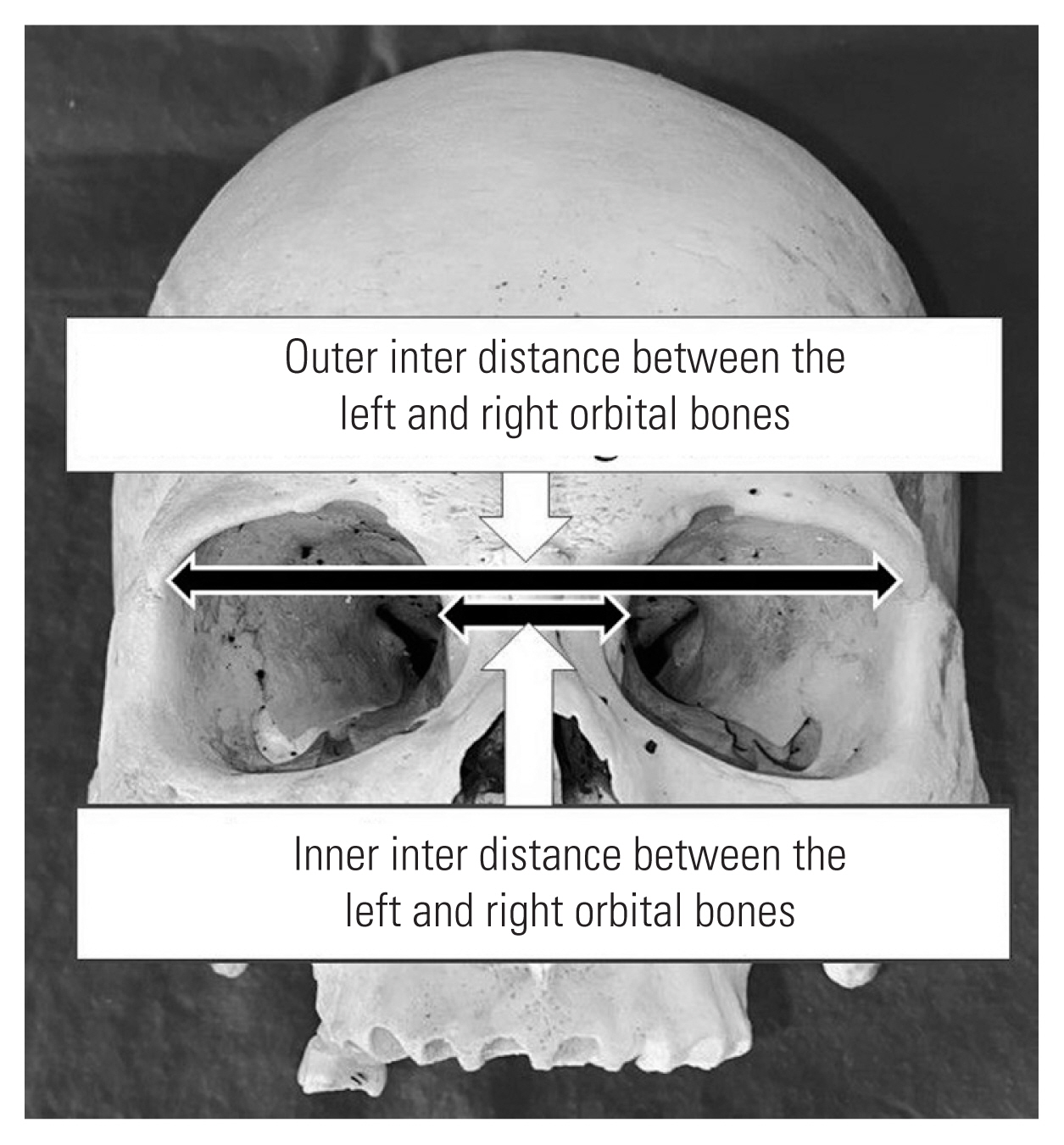
| Inner inter distances between the left and right orbital bones (mm) | 27.2±1.4e),f) | 25.7±2.6e) | 26.6±1.9 | 25.9±2.0f) | p<0.01 (p=0.0004) |
| Outer inter distances between the left and right orbital bones (mm) | 93.5±4.3g),h) | 90.0±2.8g) | 91.5±3.3 | 89.7±4.1h) | p<0.001 (p<0.00001) |
| Humeral length (cm) | 30.1±1.5 | 29.9±1.4 | 29.5±1.3 | 29.3±0.9 | NS (p=0.9059) |
| Femoral length (cm) | 42.6±1.9 | 42.1±1.9 | 41.6±2.0 | 41.1±1.5 | NS (p=0.06026) |
Values are presented as number or mean±standard deviation or number (%). In parentheses, the p-value of the Kruskal-Wallis test before the Bonferroni correction were shown. As the evaluation of the p-value after the Bonferroni correction, p<0.00833 was regarded as p<0.05, and p<0.00167 was regarded as p<0.01, and then p<0.000167 was regarded as p<0.001.
DSS, developmental spinal stenosis; DCSS, developmental cervical spinal stenosis; DLSS, developmental lumbar spinal stenosis; NS, not significant; AP, anteroposterior.
d) p<0.05 between DSS (−) group and tandem DSS (+) group in the transverse diameter of foramen magnum.
e) p<0.05 between DSS (−) group and DCSS (+) group in the inner inter distances between the left and right orbital bones.
f) p<0.05 between DSS (−) group and tandem DSS (+) group in the inner inter distances between the left and right orbital bones.
References
-
METRICS

- Related articles in ASJ
-
Biportal Endoscopic Spinal Surgery for Lumbar Spinal Stenosis2019 April;13(2)