Introduction
It is well known that extraforaminal stenosis at the lumbosacral junction causes fifth lumbar vertebral radiculopathy. The 'far-out syndrome' reported by Wiltse et al. [1] is the most well-known resultant pathological condition, which is described as an alar transverse process impingement on the spinal nerve at L5. After this first report, several other authors have reported various anatomical pathologies that produce impingement of the spinal nerve at the lumbosacral junction [2,3,4,5]. All of these conditions are clinically important and must be recognized in detail when planning an operation on a patient with fifth lumbar radiculopathy; otherwise, a lack of understanding of these conditions or inadequate treatment of this type of stenosis may cause failed back surgery syndrome [6,7]. The difficulty in detecting extraforaminal stenosis as well as foraminal stenosis at the lumbosacral junction has long been a major problem in spinal practice. The term 'hidden zone' is a metaphor used to describe the limitation of conventional imaging studies. When there is a high index of clinical suspicion, the findings of extraforaminal pathology can be guided and diagnosed using selective radiculography and/or conventional magnetic resonance imaging (MRI) [6,8]. Unrecognized extraforaminal stenosis remains one of the main causes of failed back surgery syndrome. However, the advent of three-dimensional (3D)-MRI has improved this difficulty. 3D-MRI can now be used to precisely visualize nerve structures in the hidden zone and has become a mainstay for evaluating foraminal and extraforaminal pathologies [9].
Even though extraforaminal pathology can now be recognized more easily on 3D-MRI scans, we need to pay attention to the fact that morphological abnormalities may not always reflect aspects of functional damage to the nerve such as pain generation. Using imaging studies alone has the potential risk of contributing to making a false-positive diagnosis [7] and leading to unnecessary treatment. Therefore, the establishment of techniques that can objectively examine any functional abnormality of the nerve is essential. The purpose of this study was to introduce a new effective approach to the diagnosis of extraforaminal stenosis at the lumbosacral junction using the existing electrophysiological evaluation technique.
Materials and Methods
A consecutive series of 124 patients (63 men and 61 women; mean age, 68.6 years; range, 42-88 years) with L5 radiculopathy were enrolled in this study. They underwent lumbar spine decompression surgery and intraoperative electrophysiological studies. There were 74 patients with spinal canal stenosis and 50 patients with extraforaminal stenosis at L5-S1. Their diagnoses were confirmed by various preoperative examinations of imaging studies (X-rays, computed tomography scans, MRI scans, 3D-MRI scans, myelograms, and selective nerve blocking) and by observing surgery evidence and postoperative course. On the basis of peripheral nerve conduction velocity measurements, we excluded patients with peripheral neuropathy that may interfere with nerve conduction velocity.
Each patient was placed in the prone position under general anesthesia (total intravenous anesthesia using propofol and remifentanil). Electrophysiological study was started just before surgery (skin incision). After confirmation of the patient's blood circulation and the stability of body temperature, we took preoperative selective nerve root infiltration into account and placed a pair of needle electrodes at the exit zone of the fifth lumbar nerve roots, while observing the region using fluoroscopy (Fig. 1). Another needle electrode was placed at the L2 level to serve as the anode. The L5 spinal nerve was stimulated using a standard monopolar needle placed at the exit zone of the fifth lumbar nerve roots. Recording of action potentials required a pair of surface electrodes: an active lead (G1) placed on the belly of the muscle and an indifferent lead (G2) placed on the tendon (belly-tendon recording). We then performed electrical stimulation using these electrodes and recorded compound muscle action potentials (CMAPs) from each of the tibialis anterior muscles in order to measure the distal motor latency (DML) of potentials evoked in the L5 spinal nerve. We employed measurement of the CMAPs of the tibialis anterior for examination of the L5 spinal nerve according to a past report [10].
With careful adjustment of the needle position, shocks of very low intensity elicited the maximal CMAP of the tibialis anterior. When the electrode was placed in the correct position, we were able to elicit an evoked response using weak stimulation. This enabled us to ascertain that the electrode was in the right position. An electrophysiological system (Viking IV, Nicolet Biomedical Inc., Madison, WI, USA) was used to elicit and record CMAPs via a surface electrode placed on each of the tibialis anterior muscles. The stimulus frequency was 1 Hz, the strength was 1.5-2 times the threshold level, and the duration of constant current rectangular stimulation was 0.2 ms.
The DML is defined as the time interval between the application of a stimulus and the onset of the resulting CMAP. In the presence of a localized lesion between the stimulus point and the nerve ending in the recording muscles, a conduction delay will be present (Fig. 2). This finding indicates the presence of extraforaminal entrapment neuropathy. If there is a lesion within the spinal canal only, no abnormality will be observed during conduction.
The details of this study were explained to the patients and informed consent was requested. The study was approved by the ethics committee of our university.
The data were compared between the groups by Mann-Whitney's U test. The result was considered significant if the p-value was <0.05.
Results
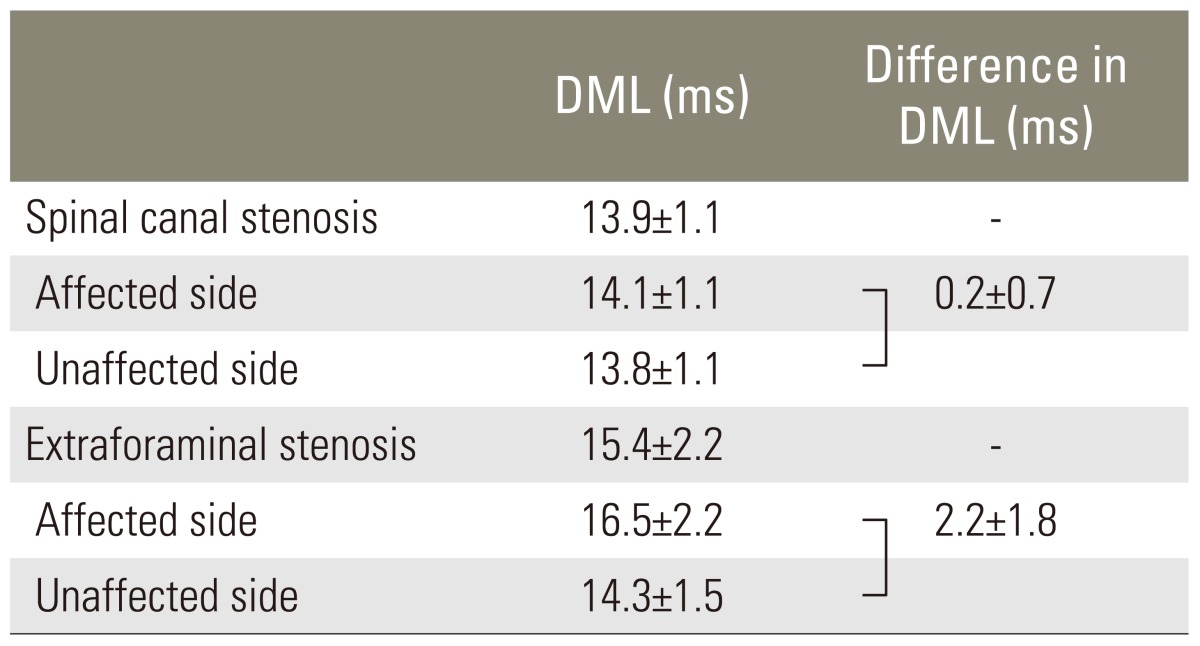
There was no problem in locating and applying a stimulus on the fifth spinal nerve. We were able to record CMAPs from the tibialis anterior muscle by a procedure that could be reliably repeated in 120 of 124 cases (96.8%). The DMLs of the potentials ranged from 10 to 24.6 milliseconds, and the laterality of latency between the 2 legs ranged from -1.5 to 8.8 milliseconds. The DMLs of the 74 patients with spinal canal stenosis ranged from 10 to 17.2 milliseconds (meanÂąstandard deviation, 13.9Âą1.1 milliseconds) (Fig. 3A), with the difference between the affected side and the unaffected side ranging from -1.5 to 3.4 milliseconds (0.2Âą0.7 milliseconds) (Fig. 3B). The DMLs of the affected side ranged from 11 to 17.2 milliseconds (14.1Âą1.1 milliseconds), and those of the unaffected side ranged from 10 to 16.4 milliseconds (13.8Âą1.1 milliseconds) (Table 1). By contrast, the DMLs of the 46 patients with extraforaminal stenosis at L5.S1 ranged from 11.2 to 24.6 milliseconds (15.4Âą2.1 milliseconds) (Fig. 4A) and the difference between the affected sides ranged from -1.0 to 8.8 milliseconds(2.2Âą1.8 milliseconds) (Fig. 4B). The DMLs of the affected side ranged from 12.8 to 24.6 milliseconds (16.5Âą2.2 milliseconds), and those of the unaffected side ranged from 11.2 to 18.5 milliseconds (14.3Âą1.5 milliseconds) (Table 1).
Statistical significant differences were noted in comparing the DML between cases with extraforaminal stenosis and those with spinal canal stenosis, as well as in comparing the differences in the DML between affected and unaffected sides in the 2 groups (p<0.01). In cases of extraforaminal stenosis, there was a statistically significant difference (p<0.01) between the DMLs of the affected side and those of the unaffected side. Using receiver operative characteristic (ROC) curve analysis, the cutoff value of the DML in the 120 patients examined was 15.2 milliseconds. The cutoff value of the difference in the DML between the affected and unaffected sides was 1.1 milliseconds.
When we employed the cutoff value for the 120 patients examined (15.2 milliseconds), the sensitivity of this electrophysiological diagnosis method was 80.4% and the specificity was 93.2% for correctly identifying extraforaminal stenosis at L5-S1. The negative predictive value was 88.5%, the positive predictive value was 88.1%, and the positive likelihood ratio was 11.8. When we employed the cutoff value of the difference between the affected and unaffected sides (1.1 milliseconds), the sensitivity of this electrophysiological diagnosis method was 76.1% and the specificity was 91.9% for correctly identifying extraforaminal stenosis at L5.S1. The negative predictive value was 86.1%, the positive predictive value was 85.4%, and the positive likelihood ratio was 9.4.
Discussion
Nerve conduction studies are well established for the diagnosis of peripheral entrapment neuropathies [11]. Evaluation of conduction provides a simple and reliable test of peripheral nerve function because diseased nerve fibers demonstrate abnormalities in conduction. In general, axonal damage results in a loss of amplitude, whereas demyelination leads to a prolongation of conduction time [10]. With this technique, we can identify any lesion and localize its location. Spinal nerves have the same fundamental anatomical structure with peripheral nerves. Hence, it is possible that the principles of nerve conduction studies can apply to the examination of extraforaminal compression neuropathies. If there is stenosis at an extraforaminal lesion and the spinal nerve is damaged at that point, propagation of a signal evoked by electrical stimulation on the lesion via a needle electrode at the extraforaminal lesion will be blocked to some degree. Therefore, there will be increased latency of CMAPs in patients with radiculopathy; we suspect that pain is generated not by axonal loss but by demyelination. As we expected, in patients with extraforaminal stenosis, the DML of the affected side was significantly longer than that of the unaffected side. This was not observed in patients with spinal canal stenosis. Therefore, laterality of the DML of the L5 spinal nerve could indicate the existence of an extraforaminal stenosis in patients with radiculopathy. We sampled the DML data and also assessed the differences in the DMLs between the affected side and the unaffected side in 120 patients. We then analyzed this data using an ROC curve to obtain the cutoff values of 15.2 milliseconds and 1.1 milliseconds, both of which may serve as a diagnostic reference measure for extraforaminal stenosis at L5-S1.
We thoroughly understand the limitations of this study, and various factors [10] that can influence nerve conduction measurements must be taken into consideration. More data are needed to standardize this method adequately. Nevertheless, to our knowledge, such a conduction study for the diagnosis of radiculopathy caused by extraforaminal stenosis has not been reported to date, while CMAPs have been widely used for the evaluation of peripheral neuropathies. As far as we know, no study attempting to assess extraforaminal pathology with this methodology has been published. The lack of recognition or inadequate treatment for extraforaminal stenosis often causes failed back surgery syndrome [6,7]. A diagnosis based on imaging studies alone has the potential risk of producing a false-positive diagnosis [7] and of leading to unnecessary treatment. Therefore, the establishment of techniques to examine any functional abnormality of the nerve is essential. The technique described in this study is straightforward to perform and the DMLs of the evoked potentials are reliable and reproducible. The technique provides a valuable diagnostic aid for patients with extraforaminal stenosis, and we believe it deserves to be introduced into popular practice.