 |
 |
- Search
| Asian Spine J > Volume 11(6); 2017 > Article |
|
Abstract
Purpose
We investigated the prevalence of neuropathic pain (NP) and patient-reported outcomes (PROs) of the quality of life (QoL) and functional disability in Korean adults with chronic low back pain (CLBP).
Methods
Patients older than 20 years with CLBP lasting for longer than three months, with a visual analog scale (VAS) pain score higher than four, and with pain medications being used for at least four weeks before enrollment were recruited from 27 general hospitals between December 2014 and May 2015. Medical chart reviews were performed to collect demographic/clinical features and diagnosis of NP (douleur neuropathique 4, DN4). The QoL (EuroQoL 5-dimension, EQ-5D; EQ-VAS) and functional disability (Quebec Back Pain Disability Scale, QBPDS) were determined through patient surveys. Multiple linear regression analyses were performed to compare PROs between the NP (DN4âĽ4) and non-NP (DN4<4) groups.
Results
A total of 1,200 patients (females: 65.7%; mean age: 63.4Âą13.0 years) were enrolled. The mean scores of EQ-5D, EQ-VAS, and QBPDS were 0.5Âą0.3, 55.7Âą19.4, and 40.4Âą21.1, respectively. Among all patients, 492 (41.0%; 95% confidence interval, 38.2%â43.8%) suffered from NP. The prevalence of NP was higher in male patients (46.8%; p<0.01), in patients who had pain based on radiological and neurological findings (59.0%; p<0.01), and in patients who had severe pain (49.0%; p<0.01). There were significant mean differences in EQ-5D (NP group vs. non-NP group: 0.4Âą0.3 vs. 0.5Âą0.3; p<0.01) and QBPDS (NP group vs. non-NP group: 45.8Âą21.2 vs. 36.3Âą20.2; p<0.01) scores. In the multiple linear regression, patients with NP showed lower EQ-5D (β=â0.1; p<0.01) and higher QBPDS (β=7.0; p<0.01) scores than those without NP.
Conclusions
NP was highly prevalent in Korean patients with CLBP. Patients with CLBP having NP had a lower QoL and more severe dysfunction than those without NP. To enhance the QoL and functional status of patients with CLBP, this study highlights the importance of appropriately diagnosing and treating NP.
Over 75% of the total population experiences at least one episode of low back pain (LBP) during their lifetime, and its annual prevalence has been reported to be 15%â45% [1]. In the Korea National Health and Nutrition Examination Survey in 2007, 15.4% of adults aged 20â89 years reported experiencing LBP, and 5.7% reported having chronic LBP (CLBP) that lasted for more than three months during the past year. An estimated 2,060,829 Koreans experienced CLBP in 2007, which is an incidence much higher than that observed in other chronic diseases [2].
Freynhagen and Baron reported that 20%â35% of patients with LBP reported accompanying neuropathic pain (NP) [3]. The International Association for the Study of Pain defined NP as pain initiated or caused by a primary lesion or dysfunction in the nervous system [4]. NP occurs when normal inflammatory responses are directed toward the body in a harmful manner [5]. The normal function of the inflammatory response is to remove damaged nerves, promote nerve regeneration, and deliver inflammatory chemokines to neuronal cell bodies, thereby stimulating proximal nociceptors and causing pain without tissue damage [6]. NP can arise from the overstimulation of pain-transmitting neuronal cell bodies and the structural alteration of synapses in the cornu of the spinal cord, interneurons, and glial cells, resulting in the development of chronic pain [5]. The characteristic clinical symptoms of NP are continuous or spontaneously provoked pain [7]. Sometimes, the pain is accompanied by sensations of burning, soreness, stinging, tingling, and hypoesthesia or dysesthesia due to neural damage [7]. Motor paralysis, cramping, and other symptoms of the autonomic nervous system have also been observed to accompany pain in damaged areas [7].
Recently, there have been various reports on the impact of chronic and refractory NP on the quality of life (QoL) and dysfunction. One review reported that NP had a negative effect on the QoL, which became more aggravated as NP became more severe [8]. Further, when NP accompanied various diseases, such as diabetes [9], spinal cord injury [10], and cancer [11], various aspects of the QoL were negatively affected. While the pain itself reduces the QoL, the dysfunction caused by this pain affects depression and the QoL [12]. In particular, CLBP causes a high absenteeism rate from work due to dysfunction; further, it shows a low cure rate and high relapse rate [13].
The early diagnosis of NP and active pain control are essential for the QoL and functional improvement of patients with CLBP. However, there has been no consensus regarding the definition of NP and its diagnostic criteria in patients with CLBP. Further, there have been no data regarding the prevalence of NP in Korean patients with CLBP, their QoL, or the level of dysfunction. Therefore, the purpose of this study was to quantify these characteristics.
This was a noninterventional, multicenter, cross-sectional observational study conducted in the orthopedic surgery and neurosurgery departments of 27 general hospitals in Korea from December 2014 to May 2015. The inclusion criteria were as follows: (1) patients aged 20 years or older; (2) patients diagnosed with LBP due to herniated disc, stenosis, spondylosis, spondylolysis, spondylolisthesis, or degenerative disc disease, according to magnetic resonance imaging or computed tomography findings; (3) patients with LBP (pain or symptoms) lasting for at least three months; (4) patients with LBP rated as âĽ4 of 10 points on a pain visual analog scale (VAS); (5) patients who received LBP-related drug therapy for over four weeks; and (6) patients who were able to understand and willing to complete the subject information sheet and informed consent form. The exclusion criteria as follows: (1) patients with LBP due to sprain, trauma, ankylosing spondylitis, myofascial pain, or sacroiliitis; (2) patients with LBP rated as <4 points on the VAS; (3) patients with LBP lasting for less than three months; (4) patients who had undergone any surgery within the past three months; (5) patients participating in another clinical study (interventional study); (6) patients with a critical or unstable health condition; or (7) patients otherwise determined unfit for inclusion or analysis by clinicians. A total of 1,200 patients who met the selection criteria were included.
A target sample size was estimated based on the assumption that the prevalence of neuropathic LBP (NLBP) is 37% [14]. With a significance level of 0.05 and an estimated error rate of 2.8%, the required number of patients to be enrolled was calculated to be approximately 1,200:
n=z21âÎą/2P (1âP)/d2 [15] (P=0.37; z21âÎą/2=1.962, when Îą is 0.05; d=0.028).
Study data were collected from patient medical records and patient surveys with a questionnaire. Medical chart reviews were conducted to collect demographic characteristics (age, sex, height, and body weight), clinical characteristics (diagnosis of LBP, comorbidities, symptom period, VAS scores, and The Quebec Task Force Classification for Spinal Disorders [QTFC-SD]), pain control state (pharmacotherapy and surgery), and diagnosis of NP. NLBP as a NP group was defined as a score of âĽ4 points on the douleur neuropathique 4 (DN4) questionnaire, which is a set of four questions on sensory descriptors and signs associated with sensory examination and has a total score range of 0 to 10 points [16]. This questionnaire is simple and includes discriminant items requiring yes or no responses. Two questions (I and II) are based on the patient's interview and the other questions (III and IV) are grounded on a standardized clinical examination [16]. QTFC-SD was mainly designed to be practical and comprised six questions. Patient classification for each question is as follows: Q1, patients who reported only back pain; Q2, patients without neurologic findings but reporting pain extend above the knee only; Q3, those with pain extending to the calf or foot; Q4, patients with pain traveling to the leg and positive neurologic findings; Q5, patients meeting criteria for Q4 and having lumbar disc disease with nerve root compression on conducting an imaging study; and Q6, those diagnosed with spinal stenosis regardless of having lumbar disc disease [17].
The QoL and degree of dysfunction were evaluated through self-administered questionnaires. The QoL was assessed with the EuroQoL 5-dimension (EQ-5D) questionnaire and EQ-VAS; higher scores imply a higher QoL in both tools (EQ-5D, â0.229â1 point; EQ-VAS, 0â100 points). The EQ-5D was composed of a total of five questions, and the scale used in the health state description part had three levels: having no problems, having some or moderate problems, and being unable to do/having extreme problems. The rated level could be coded as a number: 1, having no problems; 2, having some problems; and 3, having extreme problems. The measured EQ-5D items were converted to scores [18] using the following equation:
Final EQ-5D score [18]=1 â (0.165 + 0.003 Ă M2 + 0.274 Ă M3 + 0.058 Ă SC2 + 0.078 Ă SC3 + 0.045 Ă UA2 + 0.133 Ă UA3 + 0.048 Ă PD2 + 0.130 Ă PD3 + 0.043 Ă AD2 + 0.103 Ă AD3 + 0.347 Ă N3 + 0.014 Ă I2sq)
The degree of dysfunction was measured using the Quebec Back Pain Disability Scale (QBPDS), which is composed of 20 questions with a scale of 0 to 5 for each question and a total score ranging from 0 to 100 points. A higher score indicates more severe dysfunction (for more information about the QBPDS, see the footnote in Fig. 1).
The patients (or their legal representatives) were provided with all study-related information, and they signed an informed consent form. All participating medical institutions obtained approval from their respective institutional review boards.
Patient demographics and clinical characteristics are summarized as meanÂąstandard deviation for continuous variables or frequency (percentage) for categorical variables. Prevalence rates of NP by patient characteristics were compared using the Ď2 test, while the QoL and degree of dysfunction between the NP and non-NP groups were compared using Student's t-test. To examine whether NP is associated with either QoL or the severity of dysfunction, multiple linear regression analysis was performed with adjustment for effects of potential confounders. Variables with p-values of <0.1 in bivariate analyses were selected for adjustment in the multivariable model. All statistical analyses were performed using the SAS software, version 9.4 (SAS Institute, Inc., Cary, NC, USA), and two-tailed p-values less than 0.05 were considered statistically significant.
The mean age of the patients was 63.4Âą13.0 years. Most patients (65.7%) were females and 702 (58.5%) had one or more comorbidities. The most common comorbidity was hypertension (44.1%); this was followed by diabetes (19.5%). Rheumatoid arthritis (3.2%) was the least frequent comorbidity. The mean pain VAS score in patients with CLBP was 6 points. The mean duration of pain was four years and two months, and the mean duration of disease after diagnosis was one year and five months. The most frequently diagnosed condition was stenosis (47%), and the most frequent pain type identified by the QTFC-SD was pain with distal extremity radiation (40%; Table 1).
The mean DN4 score was 3.4Âą2.0 points. Among the subitems, the most common response was âpain related with tingling symptoms in the same area (22.4%)â (Fig. 2).
The NP prevalence was 41% (95% confidence interval, 38.2%â43.8%). NP was more prevalent in males. Among the QTFC-SD items, the proportions of patients with radiating pain and NP were higher than those of patients with other types of pain. Patients with severe pain (VAS scores, 7â10) had a higher prevalence of NP than those with moderate pain (VAS scores, 4â6) (Table 2).
The mean EQ-5D, EQ-VAS, and QBPDS scores in patients with CLBP were 0.5Âą0.3, 55.Âą19.4, and 40.4Âą21.1 points, respectively. Between the NP and non-NP groups, significant mean differences were found in the EQ-5D (NP group vs. non-NP group: 0.4Âą0.3 vs. 0.5Âą0.3; p<0.01) and QBPDS (NP group vs. non-NP group: 45.8Âą21.2 vs. 36.3Âą20.2; p<0.01) scores. No significant mean difference in the EQ-VAS score between the two groups was shown (NP group vs. non-NP group: 54.7Âą20.0 vs. 56.7Âą18.9; p=0.07) (Fig. 3). The percentages of patients with NP who answered level 3 (a lot of problems) in each dimension of EQ-5D were pain/discomfort (36.2%), anxiety/depression (10.7%), usual activities (10.5%), mobility (5.0%), and self-care (4.1%) (Fig. 1A). In the NP group, the proportion of patients who responded âI was very uncomfortable or unable to do any activity in each item of the QBPDSâ was the highest in item 12: Run one block (approximately 100 m; 15.0%) followed by item 20, moving heavy travel luggage (56.2%). Item 13, taking out foods from the refrigerator (7.3%), showed the lowest proportion. The responses to the QBPDS questionnaire are summarized in Fig. 1B.
After adjustment with confounding variables, the NP group had significantly lower EQ-5D (β=â0.1; p<0.01) and significantly higher QBPDS (β=7.0; p<0.01) scores than the non-NP group. While there was no significant difference in the EQ-VAS scores between the two groups, the EQ-VAS score was lower in the NP group than in the non-NP group (β=â2.6; p=0.059) (Table 3).
This study revealed a high NP prevalence (41.0%) based on the DN4 questionnaire in Korean patients with CLBP. Similar results have been identified in other studies. In Turkey, NP prevalence in patients with CLBP was 39.4% in 2014 as measured by the Leeds Assessment of Neuropathic Symptoms and Signs (LANSS) [19], and a multicenter study in Saudi Arabia found a prevalence of 54.7%, also measured by the LANSS [20]. In a multicenter Japanese study of patients with spinal disorder having chronic pain, the prevalence of NP was 53.3%, as assessed by an NP screening questionnaire [21]; this study included assessments of lumbar/sacral and cervical levels. If cervical and thoracic levels were also considered for our patients, a higher NLBP prevalence would have been obtained. An American study using the PharMetrics IMS LifeLinkTM US Claims Database (2006â2008) found an NLBP prevalence of 90.4% based on ICD-9 codes, indicating that almost all patients with CLBP had NP [22]. Given the substantial differences in the methods of diagnosis for NP, direct comparisons with the prevalence of NLBP reported in other studies are limited.
Numerous factors increased NP prevalence in patients with CLBP. There was a higher prevalence of NP in males in the present study. This is because men tended to spend a longer time at work [23]. The prevalence of NP in the young population was higher than in patients aged 65 years and older, which is similar to the findings of Sakai et al. [24] Inflammation and chronic obstructive pulmonary disease, in particular, increased NP prevalence, suggesting that patients with low immunity or abnormal immune status are more prone to NP [25]. Regarding the primary diagnosis, NP was common in patients with spondylolysis, herniated disc, and degenerative disc disease, which suggests that diseases that directly damage the nerves have a higher NP incidence. Of the types of pain assessed by the QTFC-SD, there was a higher prevalence of NP in patients who reported radiating pain. Further, it seems that items in the QTFC-SD that are related to pain in the lower limbs are more helpful for accurately diagnosing NP than items related to lower back pain [26]. As pain became more severe, NP prevalence increased, which was consistent with the results of a previous study [27]. Finally, as the disease duration increased, the severity of pain also increased, suggesting that early detection and treatment are vital for managing LBP.
In a study on the degree of dysfunction in 1,760 patients with CLBP using the QBPDS [28], patients with CLBP had severe dysfunction with a mean score of 51.7Âą15.6 points, which was similar to the degree of dysfunction in the NLBP patients in our study. NP caused by a lesion or dysfunction affecting the nervous system [4] can increase pain and reduce physical activity [1929].
Korean patients with CLBP had a much lower QoL than patients with other chronic diseases [18]. According to our study, patients with CLBP and NP have an even worse QoL than those without NP. Recent studies have reported that NP can adversely affect patients' overall QoL [30] because NP hampers functional movements and amplifies depression, which interferes with proper self-management and daily life [8]. It is reasonable to conclude that the combination of NP and CLBP leads to poorer outcomes than either condition by itself. Further, failures in early pain control measures or a lack of targeted treatment might have exacerbated outcomes in our patients.
Therefore, to administer the appropriate treatment and motor rehabilitation regimen for each individual that ultimately will restore normal functionality and improve the QoL, the underlying NP mechanism in patients with NLBP must be identified.
A large-scale epidemiologic study of 1,200 patients from 27 general hospitals was conducted to investigate NP prevalence in adults with CLBP in Korea. To our knowledge, there have been no other similar studies conducted in a Korean population. The results of the QoL and degree of dysfunction surveys suggest that patients with CLBP should be screened for NP and that the choice of therapy should be based on the underlying NP mechanism.
There are several limitations. One is various disease categories that we included, such as LBP and radiculopathy, which might have confounded the results. Further, because this is a cross-sectional study, we were unable to evaluate the temporal and causal relationships between patient characteristics and NP prevalence.
NP was highly prevalent in Korean patients with CLBP. In particular, NP prevalence was higher in males and in patients who had radiating and severe pain. In addition, patients with CLBP having NP had a lower QoL and more severe dysfunction than those without NP. Therefore, patients with such characteristics should be carefully examined for NP, and the underlying mechanism of NP should be identified to administer the appropriate treatment for NLBP.
Acknowledgments
This research was sponsored by Pfizer Pharmaceuticals Korea Ltd. J. H. Kim and J. T. Hong equally contributed as a first author
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
1. Shin SW. Low back pain: review of anatomy and pathophysiology. J Korean Med Assoc 2006;49:656â664.

2. Jhun HJ, Park JY. Estimated number of korean adults with back pain and population-based associated factors of back pain: data from the fourth Korea national health and nutrition examination survey. J Korean Neurosurg Soc 2009;46:443â450. PMID: 20041054.



3. Freynhagen R, Baron R. The evaluation of neuropathic components in low back pain. Curr Pain Headache Rep 2009;13:185â190. PMID: 19457278.


4. Merskey H, Bogduk N. Classification of chronic pain: descriptions of chronnic pain syndromes and definitions of pain terms. 2nd ed. Seattle: IASP Press; 1994.
5. Ki MY, Kim SM. Diagnosis and Treatment of Neuropathic Pain. J Korean Med Assoc 2008;51:1139â1148.

6. Malin SA, Molliver DC, Koerber HR, et al. Glial cell line-derived neurotrophic factor family members sensitize nociceptors in vitro and produce thermal hyperalgesia in vivo. J Neurosci 2006;26:8588â8599. PMID: 16914685.



7. Ko Y, Kim YH. The pharmacological management of neuropathic pain. J Korean Med Assoc 2012;55:582â592.

8. Jensen MP, Chodroff MJ, Dworkin RH. The impact of neuropathic pain on health-related quality of life: review and implications. Neurology 2007;68:1178â1182. PMID: 17420400.


9. Dermanovic Dobrota V, Hrabac P, Skegro D, et al. The impact of neuropathic pain and other comorbidities on the quality of life in patients with diabetes. Health Qual Life Outcomes 2014;12:171PMID: 25468384.



10. Aquarone RL, Faro ACMe, Nogueira PC. Central neuropathic pain: implications on quality of life of spinal cord injury patients. Rev Dor 2015;16:280â284.

11. Lema MJ, Foley KM, Hausheer FH. Types and epidemiology of cancer-related neuropathic pain: the intersection of cancer pain and neuropathic pain. Oncologist 2010;15(Suppl 2): 3â8.


12. Park KS, Ryoo EN, Choi MH. The effect of balance taping therapy on pain of the lower back pain patient. J Korean Acad Adult Nurs 2005;17:77â87.
13. Chung SG. Rehabilitative Treatments of Chronic Low Back Pain. J Korean Med Assoc 2007;50:494â506.

14. Freynhagen R, Baron R, Gockel U, Tolle TR. pain-DETECT: a new screening questionnaire to identify neuropathic components in patients with back pain. Curr Med Res Opin 2006;22:1911â1920. PMID: 17022849.


15. Aday LA, Cornelius LJ. Designing and conducting health surveys a comprehensive guide. San Francisco: John Wiley; 2006.
16. Bouhassira D, Attal N, Alchaar H, et al. Comparison of pain syndromes associated with nervous or somatic lesions and development of a new neuropathic pain diagnostic questionnaire (DN4). Pain 2005;114:29â36. PMID: 15733628.


17. Atlas SJ, Deyo RA, Patrick DL, Convery K, Keller RB, Singer DE. The Quebec Task Force classification for Spinal Disorders and the severity, treatment, and outcomes of sciatica and lumbar spinal stenosis. Spine (Phila Pa 1976) 1996;21:2885â2892. PMID: 9112713.


18. Korea Institute for Health and Social Affairs (KIHASA). Valuing Health States Using EQ-5D Final Report. Sejong: KIHASA; 2005.
19. ĂalÄąk Y, Filiz ĂalÄąk A. The evaluation of the effect of neuropathic pain on functional disability in patients with chronic low back pain. Turk J Osteoporos 2015;21:122â126.

20. Kaki AM, El-Yaski AZ, Youseif E. Identifying neuropathic pain among patients with chronic low-back pain: use of the Leeds Assessment of Neuropathic Symptoms and Signs pain scale. Reg Anesth Pain Med 2005;30:422â428. PMID: 16135345.


21. Yamashita T, Takahashi K, Yonenobu K, Kikuchi S. Prevalence of neuropathic pain in cases with chronic pain related to spinal disorders. J Orthop Sci 2014;19:15â21. PMID: 24306580.


22. Mehra M, Hill K, Nicholl D, Schadrack J. The burden of chronic low back pain with and without a neuropathic component: a healthcare resource use and cost analysis. J Med Econ 2012;15:245â252. PMID: 22136441.


23. Purwata TE, Sadeli HA, Yudiyanta , et al. Characteristics of neuropathic pain in Indonesia: a hospital based national clinical survey. Neurol Asia 2015;20:389â394.
24. Sakai Y, Ito K, Hida T, Ito S, Harada A. Neuropathic pain in elderly patients with chronic low back pain- and effects of pregabalin: a preliminary study. Asian Spine J 2015;9:254â262. PMID: 25901238.



25. Austin PJ, Moalem-Taylor G. The neuro-immune balance in neuropathic pain: involvement of inflammatory immune cells, immune-like glial cells and cytokines. J Neuroimmunol 2010;229:26â50. PMID: 20870295.


26. Attal N, Perrot S, Fermanian J, Bouhassira D. The neuropathic components of chronic low back pain: a prospective multicenter study using the DN4 Questionnaire. J Pain 2011;12:1080â1087. PMID: 21783428.


27. Torrance N, Smith BH, Watson MC, Bennett MI. Medication and treatment use in primary care patients with chronic pain of predominantly neuropathic origin. Fam Pract 2007;24:481â485. PMID: 17670804.


28. Verkerk K, Luijsterburg PA, Heymans MW, et al. Prognosis and course of disability in patients with chronic nonspecific low back pain: a 5- and 12-month follow-up cohort study. Phys Ther 2013;93:1603â1614. PMID: 23824781.


29. Foley KM. Opioids and chronic neuropathic pain. N Engl J Med 2003;348:1279â1281. PMID: 12660393.


30. Oster G, Harding G, Dukes E, Edelsberg J, Cleary PD. Pain, medication use, and health-related quality of life in older persons with postherpetic neuralgia: results from a population-based survey. J Pain 2005;6:356â363. PMID: 15943957.


Fig. 1
Differences between the NP and non-NP groups on the quality of life and level of functional disability. (A) Proportion of Level 3 (a lot of problems) in response to each EQ-5D dimension. (B) Proportion of level 4 and 5 (very difficult & unable to do) in response to each QBPDS item. NP, neuropathic pain; EQ-5D, Euro quality of life 5-dimension; QBPDS, Quebec Back Pain 6 Disability Scale; Item 1, get out of bed; Item 2, sleep through the night; Item 3, turn 7 over in bed; Item 4, ride in a car; Item 5, stand up for 20â30 minutes; Item 6, sit in a chair for several hours; Item 7, climb one flight of stairs; Item 8. walk a few blocks (300â400 m); Item 9, walk several kilometers; Item 10, reach up to high shelves; Item 11, throw a ball; Item 12, run one block (about 100 m); Item 13, take food 10 out of the refrigerator; Item 14, make your bed; Item 15, put on socks (pantyhose); Item 16, bend over to clean 11 the bathtub; Item 17, move a chair; Item 18, pull or push heavy doors; Item 19, carry two bags of groceries; Item 20, lift and carry a heavy suitcase. a)p<0.01; b)p<0.05.

Fig. 2
Neuropathic pain in patients with chronic low back pain (n=1,200). DN4, douleur neuropathique 4; SD, standard deviation. a)Multiple responses item.

Fig. 3
Quality of life and functional disability in patients with chronic low back pain. (A) EQ-5D. (B) EQ-VAS. (C) QBPDS. NP, neuropathic pain; PRO, patient reported outcome; EQ-5D, Euro quality of life 5-dimension; EQ-VAS, Euro quality of life visual analog scale; QBPDS, quebec back pain disability scale. a)p-value by Student's t-test; b)Values given as meanÂąstandard deviation.

Table 3
Quality of life and functional disability in chronic low back pain with neuropathic pain
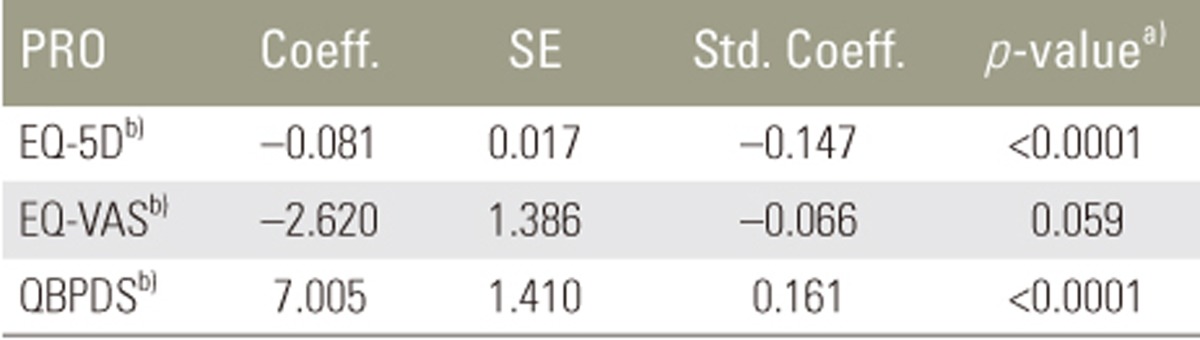
PRO, patient reported outcome; Coeff., coefficient; SE, standard error; Std. Coeff., standardized coefficient; EQ-5D, Euro quality of life 5-dimension; EQ-VAS, Euro quality of life visual analog scale; QBPDS, quebec back pain disability scale.
a)p-value by multiple linear regression analysis; b)Reference group: non-neuropathic pain patients; adjusted variables were sex, age, Quebec Task Force Classification for Spinal Disorders, duration of LBP after diagnosis, pain visual analog scale.








