|
 |
- Search
| Asian Spine J > Volume 17(5); 2023 > Article |
|
Abstract
Study Design
A retrospective comparative study of denosumab therapy adherence before and during the coronavirus disease 2019 (COVID-19) pandemic.
Overview of Literature
Denosumab is a monoclonal antibody for osteoporosis treatment. Delayed denosumab injections are associated with reduced treatment responses, which was a concern during the COVID-19 pandemic.
Methods
Participants included 376 patients who received denosumab (60 mg every 6 months) from January 2013 to June 2021. The time from therapy initiation to discontinuation was used to measure persistence, and that between initial and subsequent injections was used to determine adherence. The pandemic period was from March 2020 to December 2021.
Results
Patients were divided into those treated after March 2020 (pandemic group, n=244) and those who discontinued treatment before March 2020 (non-pandemic group, n=132). Non-persistent cases accounted for 154, including 24 (20%), 64 (19%), and 66 (53%) aged ≤59 years, 60–79 years, and ≥80 years, respectively. The overall persistence rate at 78 months was 59.2%. Postponed cases were significantly lower in the non-pandemic group than in the pandemic group (8% vs. 15%, p=0.042). Postponement with a delay of 1–2 months did not significantly differ between the two groups, but with a significant difference for a delay of ≥3 months (0% vs. 36%, p=0.024).
Osteoporosis has become a major public health concern with the aging of the population [1–3]. The pathology includes reduced bone mass and microarchitecture deterioration of bone tissue, and osteoporosis manifests as a chronic disease that causes pain and disability [4,5]. Osteoporosis and fragility fractures that occur as complications result in major economic costs [6]. Several drugs are used for treatment, but these drugs often have poor adherence. This limits the therapeutic benefit and increases the economic costs, and adherence is an important element in international guidelines for osteoporosis treatment [7].
Denosumab (Prolia; Amgen, Thousand Oaks, CA, USA) is a human monoclonal antibody that selectively binds to the RANK ligand and has an antiresorptive effect in osteoporosis [8]. Additionally, denosumab is the first biological agent for osteoporosis. It is administered at 6-month intervals by subcutaneous injection and has been authorized for osteoporosis in Japan since 2013 [9], with a requirement of administration by nurses or physicians in hospital clinics or in private specialist and general practitioner clinics because of the risk of an anaphylactic reaction. Delayed denosumab injections have been associated with suboptimal bone mineral density (BMD) responses in the spine and total hip [10]. Missed or delayed denosumab doses may be associated with loss of gained BMD and rebound vertebral fractures [11]. This increased risk of vertebral fractures may occur as early as 4–8 weeks after injection schedule interruption [12], and therefore, excessive delays in injections should be avoided. This concern may explain the consistently higher denosumab adherence than that for other anti-osteoporotic agents [13,14].
The coronavirus disease 2019 (COVID-19) pandemic has greatly affected healthcare worldwide, despite the many measures taken to contain the virus. The continuation of anti-osteoporosis treatment is necessary during the pandemic, but the impact of COVID-19 on denosumab treatment continuation and adherence in a real-world setting in Japan is unknown. This is particularly important because deleterious effects may occur in the first few months after denosumab discontinuation, including accelerated bone turnover, rapid BMD loss, and increased multiple vertebral fractures [15,16]. Thus, patients need to receive denosumab treatment on time. Here, we report our experience of denosumab adherence and postponed treatment before and during the COVID-19 pandemic.
This retrospective study reviewed charts of patients treated subcutaneously with denosumab (60 mg every 6 months). The inclusion criteria were as follows: treatment for primary osteoporosis, bone density of ≤70% of the young adult mean (YAM), proximal femoral or vertebral fracture in medical history, and fragile fractures with a bone density of ≤80% of YAM. Cases of secondary osteoporosis caused by steroid use were also included. The referring specialist provided each patient with instructions on the use of denosumab at the start of treatment. Information was collected for age, period of use, fracture history, glucocorticoid use, rheumatoid arthritis, use of concomitant vitamin D or active vitamin D3, year of initiation, cause of non-persistence, postponed treatment, and period of postponement. Persistence was defined as the duration from initiation to discontinuation of therapy [17] and evaluated as a dichotomous variable at the end of a predefined period, with cases defined as “persistent” or “non-persistent.” Persistence rates were calculated for all eligible patients [18], and Kaplan-Meier curves were used to depict persistence over time in each age group. The period between the initial and subsequent denosumab injections was used as a measure of compliance regarding adherence: cases were defined as compliance if this period was 6 months, whereas those in which the drug was delayed for >1 month from the scheduled administration time were defined as postponed. The study was approved by the Institutional Review Board of Japan Red Cross Aichi Medical Center, Nagoya Daini Hospital (IRB approval no., 506-9019), and informed consent was obtained from all patients.
The Kaplan-Meier analysis was used to estimate the persistence rate. Continuous and categorical variables are expressed as mean±standard deviation and as the absolute value and percentage, respectively. Student t-test and Kruskal-Wallis test were used to analyze the differences between two and three groups, respectively. All calculations were performed using IBM SPSS ver. 24.0 (IBM Corp., Armonk, NY, USA), with p-values of <0.05 considered significant in all analyses.
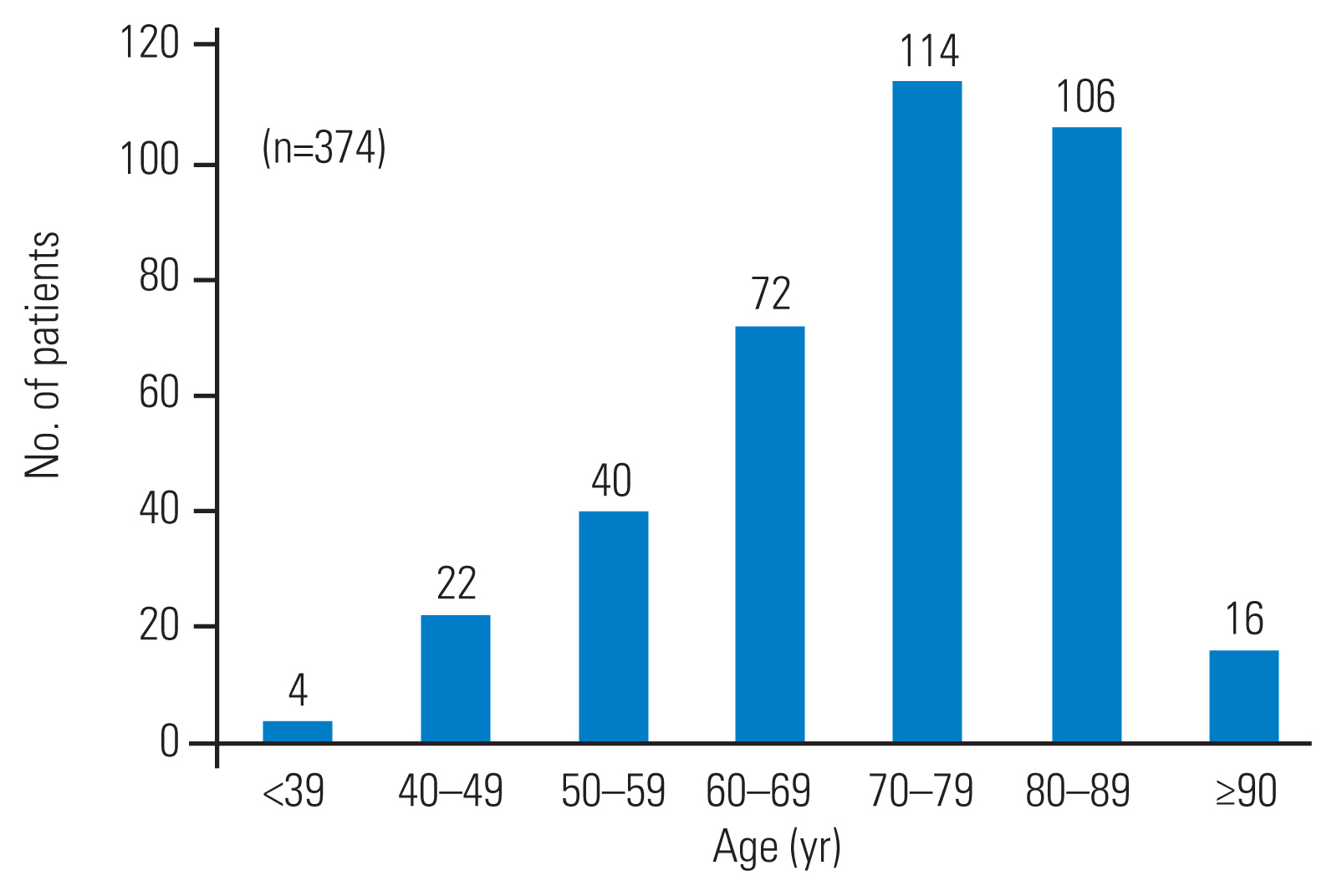
The study included 376 patients who started denosumab for osteoporosis from January 2013 to June 2021. The COVID-19 pandemic was from March 2020 to December 2021. Patients who received treatment after March 2020 were included in the pandemic group (n=244), whereas patients who discontinued treatment before March 2020 were defined as the non-pandemic group (n=132). Fig. 1 shows the flow diagram for the study population. Fig. 2 shows the age distribution of patients. Four patients were <40 years old and 16 were >90 years old, indicating a wide age range. The average age was 71.8±13.3 years (range, 35–93 years). The median follow-up period was 25.7±18.9 months (range, 6.0–86.1 months).
Table 1 shows the background and characteristics of patients in the non-pandemic and pandemic groups. The postponement rate was significantly higher in the pandemic group (8% versus 15%, p=0.042). The postponement rate did not significantly differ with a delay between 1 and 2 months but significantly differed for a delay of ≥3 months between the groups (0% versus 36%, p=0.024) (Table 1). Non-persistent cases accounted for 154, including 24 (20%), 64 (19%), and 66 (53%) aged ≤59 years, 60–79 years, and ≥80 years, respectively. Non-persistence was caused by transfer to another hospital (n=49), change to another drug (n=47), outpatient visit interruption (n=34), dental treatment (n=12), adverse events (n=4), and patient request (n=8) (Table 2). Of the 47 patients who switched to another drug, 12 were during the pandemic period, including four cases that switched to an oral bisphosphonate. The adverse events were fatigue, skin rash, and nausea (not shown in Table 2). Table 3 shows the reasons for administration postponement. A medical examination was not performed on 22 patients (61%) of the pandemic group due to a request for self-restraint by the government. Additionally, four patients were infected with COVID-19, and thus could not be seen. Infection-related deaths were not observed, as well as postponements due to logistic reasons (Table 3).
The overall persistence rate at 78 months was 59.2%, and the Kaplan-Meier analysis revealed a declined persistence to 84.2%, 80.1%, 76.3%, 73.1%, and 69.1% at 12, 24, 36, 48, and 60 months, respectively (Fig. 3A). Persistence was significantly lower in patients aged ≥80 years than in those aged <60 years and 60–79 years (both p<0.01) (Fig. 3B). A significant difference was found in age at initiation of denosumab between non-persistent (n=154) and persistent (n=222) cases (76.5 years versus 68.5 years, p<0.01) (Fig. 4). Of the 154 non-persistence cases, 132 occurred during the non-pandemic period (January 2013–February 2020) and 22 during the pandemic period (March 2020–December 2022), giving an incidence of non-persistence cases of 16.1 and 12.9 cases/year in the respective periods (Table 4). The pandemic group indicated a significantly longer follow-up period for postponed cases than non-postponed cases (35.7 months versus 29.6 months, p=0.025) (Table 5).
The COVID-19 pandemic disrupted the provision of health care in many countries. Measures aimed at containing the spread of the virus, including lockdowns, may have negatively affected patients with both acute and chronic diseases. However, the lockdown was not implemented as a national policy in Japan. The present study examined adherence to treatment with denosumab injections before and during the COVID-19 pandemic, using data from all departments at a single center. These data reflect the situation at an affiliated emergency hospital in Japan, which include young to elderly patients. Moreover, this is the first report on denosumab adherence during the COVID-19 pandemic in Japan.
Denosumab is a fully human monoclonal antibody that is approved for osteoporosis treatment and slows disease progression through binding to the RANKL receptor activator, with the results of increased BMD and reduced fracture risk. Denosumab has beneficial pharmacological effects, but treatment adherence based on clinical guidelines is critical for the efficacy of denosumab in osteoporosis treatment. This emphasizes the importance of identifying the root causes of poor adherence and working towards improving this aspect of clinical practice. Patients with more information and improved motivation for osteoporosis treatment may be more adherent, and clinical trials show high persistence with denosumab [19]. A multicenter study of 935 postmenopausal women treated for osteoporosis in the United States and Canadian community practice by Silverman et al. [20] revealed a 12-month discontinuation rate of 18%. Migliaccio et al. [18] revealed persistence rates of 83%, 69%, and 62% at 12 months, 18 months, and 24 months, respectively. An analysis of 2,315 cases in the Swedish Prescribed Drug Register (n=2,315) by Karlsson et al. [21] revealed denosumab persistence of 83% and 62% at 12 months and 24 months, respectively, in real-world data.
Persistence is commonly considered important for chronic disorder management, and better adherence and persistence may result from longer dosing intervals [22]. Understanding the factors that affect perceptions of osteoporosis treatment among patients may increase adherence through improved patient education. Persistence is better for injectable anti-osteoporotic drugs, such as teriparatide and denosumab, and key factors for improving persistence include appropriate patient monitoring, visit frequency, pharmacological schedule, and patient motivation; all of these may be important in increasing the effectiveness of anti-osteoporotic therapy [17].
The current study revealed persistence rates for denosumab of 84.2% and 69.1% at 12 months and 60 months, respectively. These rates are not as high as in previous reports, which may be because some of our cases had forced interruption of dosing to permit dental treatment for jaw bone necrosis prevention. Some doctors recommend denosumab withdrawal in dentistry, especially for tooth extraction, and several such cases were defined as non-persistent. The transfer to another hospital to visit a local doctor also contributed to non-persistence, and this was particularly important in our patients, many of whom were over 90 years old. Such patients might have difficulty traveling to a hospital and may prefer local medical treatment. Additionally, transfer to another hospital does not preclude denosumab continuation, and the inclusion of such cases would increase the persistence rate. Some patients also discontinued treatment at outpatient clinics, including deaths in elderly patients due to their shorter life expectancy. Non-persistence occurred due to adverse events, such as fatigue and nausea, and doctors forget to order the drug in a few cases. Thus, efforts of medical personnel for promoting continuation are also necessary.
Some studies using surveys have suggested a negative impact of the COVID-19 pandemic on osteoporosis care [23,24]. Patients appeared to be hesitant to come to hospitals and clinics for the management of chronic conditions, including osteoporosis, during the pandemic period in Japan. Thus, factors associated with the use of denosumab and those related to patients may explain the lower compliance rates. First, denosumab is only available through subcutaneous injection administered by medical professionals, and concerns about the safety of the hospital environment may have affected the willingness of patients to receive injections during the pandemic period. Second, physicians have a significant impact on denosumab adherence because they can set the dosing schedule, track regularity, and convey the importance of compliance to the patient when the injection is delayed. Therefore, the Japan Osteoporosis Society provided evidence-based clinical recommendations to counteract difficulties with osteoporosis treatment as a measure against non-adherence during the pandemic period [25]. The panel of experts recommended a “temporary transition to an oral bisphosphonate for patients in who continued treatment with denosumab is not feasible within 6 months of their most recent prior denosumab injection.” Our series revealed that 12 patients switched to another drug during the pandemic period, including four cases switching to an oral bisphosphonate. These results might have been influenced by recommendations from the Osteoporosis Society. However, our series included 13 cases with a postponement of ≥3 months during the pandemic period that did not require a drug change in the recommendations. These recommendations should be considered as long as the COVID-19 pandemic continues.
Denosumab is subcutaneously administered once every 6 months, and this results in higher adherence compared to that for other agents [26]. Adherence is significantly lower in patients aged ≥80 years [27], but these findings did not include the effects of the COVID-19 pandemic. Chandran et al. [28] revealed significantly reduced adherence to the 6-monthly dosing schedule of denosumab during the pandemic lockdown period, compared to that in pre-COVID-19 times. Minisola et al. [29] revealed a marked increase in the number of patients with vertebral fractures after denosumab discontinuation due to the inability to access medical facilities during the pandemic. The COVID-19 pandemic was characterized by patients avoiding visits to medical facilities, but with no lockdown in Japan. Thus, non-persistence cases were not particularly common during the pandemic in our series, although some cases of denosumab postponement or discontinuation were observed. Our findings are of note as the first English-language report of denosumab adherence during the COVID-19 pandemic in Japan.
Non-persistence associated with the pandemic period did not increase in our series. However, postponed cases were significantly more during the pandemic period, especially with delays of ≥3 months. Additionally, postponed cases had a significantly longer follow-up period, which suggests that the physician or the patient themselves fully understood the importance of regular denosumab administration in the COVID-19 pandemic environment. Conversely, the possibility of infection may have caused hesitancy regarding a hospital visit. Huang et al. [22] suggested that waiting for >9 months between denosumab injections should be avoided because circulating drug levels steadily decline at 6 months after administration, with the drug being eliminated at 9 months. Additionally, bone turnover markers generally reach pre-treatment levels within 9 months after the last denosumab dose [30]. The current study findings suggest that clinicians may have some flexibility to prescribe denosumab, particularly in circumstances, such as the COVID-19 pandemic, but the time between injections should not be excessively extended.
The study has several limitations. First, this is a retrospective small series in a single center. Second, we did not evaluate BMD as a therapeutic effect measure. Third, the study focused on patients receiving denosumab alone and lacks a comparator group of patients receiving a different drug. Fourth, the primary aim was to evaluate the adherence of patients during the COVID-19 pandemic, and not to evaluate adherence after the interruption of denosumab therapy. However, this is the first English-language report including males and females of all ages taking denosumab during the COVID-19 pandemic in Japan. The data made it possible to focus on the cause of denosumab discontinuation. Several limitations causing underestimation of adherence have been identified in prescription-based databases. However, denosumab is injected subcutaneously by hospital staff in Japan, and this ensures reliable follow-up data.
This study focused on the cause of denosumab non-persistence during the COVID-19 pandemic in Japan. The results indicated a generally high persistence rate, with lower persistence in elderly patients. Denosumab adherence rates did not decrease during the pandemic but with a significant increase in postponed cases. Therefore, reasons for non-adherence in such situations should be further studied. Better communication by health providers regarding the critical need for adherence to agents, such as denosumab, and alternatives for delivery and safe administration are required to avoid dosing schedule interruptions. These approaches could promote treatment in super-aging populations, including in Japan, even under future pandemic conditions.
Notes
Author Contributions
KK analyzed data and wrote the manuscript, KS designed the study and collected data, TA designed the study and collected data, KA designed the study and collected data, and SI designed the study and collected data. All authors have read and agreed to the published version of this manuscript.
Fig. 1
(A) Flow diagram for patients (n=376) in the pandemic (n=244) and non-pandemic (n=132) groups. (B) Definition of the pandemic period (March 2020 to December 2021) and the non-pandemic period (January 2013 to February 2020).

Fig. 3
(A) Kaplan-Meier plot showing the probability of all patients continuing with denosumab treatment at a given time point. The rate dropped to 84.2%, 80.1%, 76.3%, 73.1%, and 69.1% at 12, 24, 36, 48, and 60 months of follow-up, respectively. (B) Kaplan-Meier plot showing the probability of patients in each age group continuing with denosumab treatment at a given time point. Persistence was significantly lower in patients aged ≥80 years than in those aged ≤59 years and 60–79 years (both p<0.01).
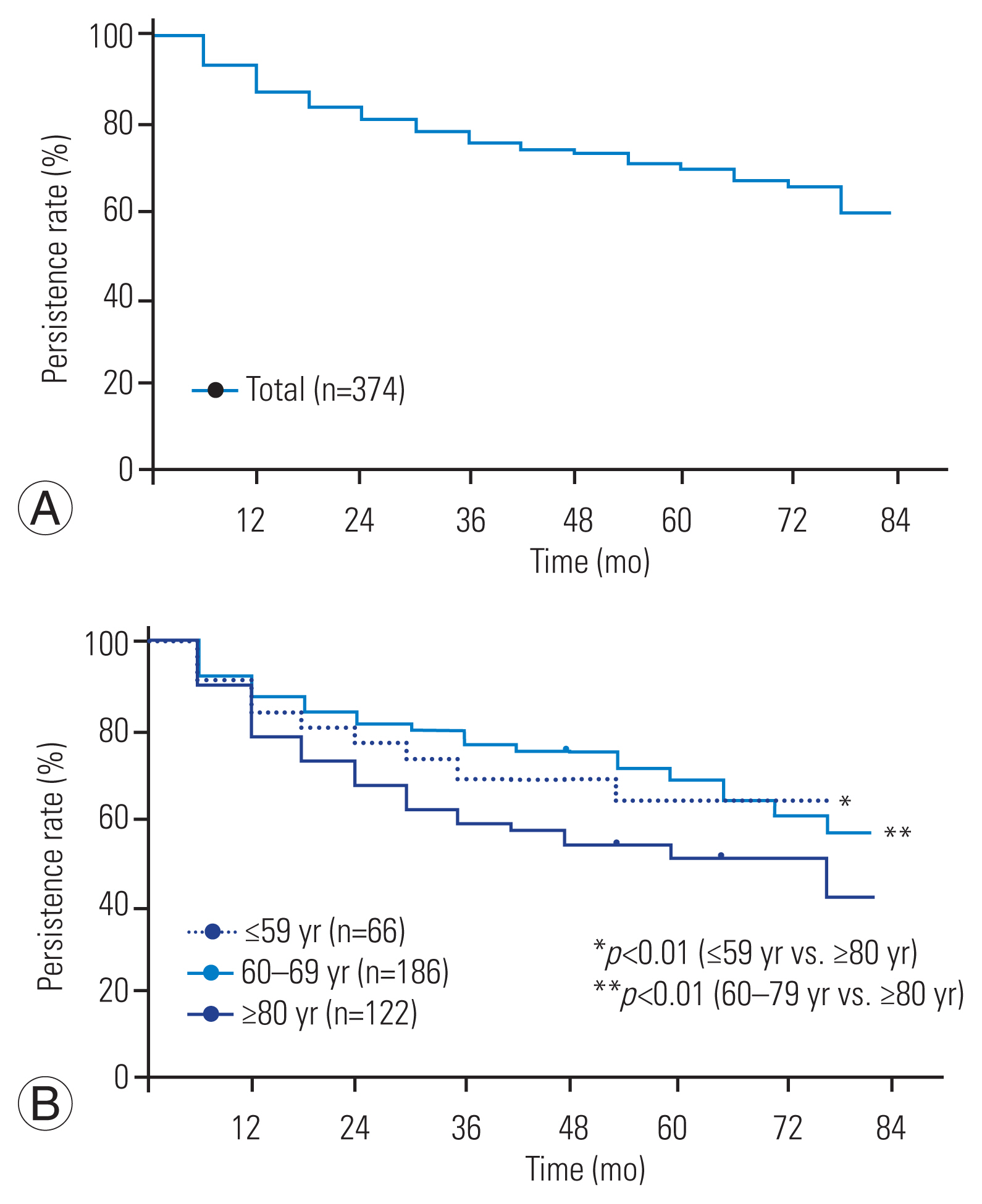
Fig. 4
Age at initiation of denosumab treatment in non-persistence (n=154) and persistence (n=222) cases. Age was significantly higher in non-persistence cases (76.5 years vs. 68.5 years, p<0.01).

Table 1
Characteristics of patients in the non-pandemic and pandemic groups
| Characteristic | Total (n=376) | Non-pandemic (n=132) | Pandemic (n=244) | p-value |
|---|---|---|---|---|
| Age (yr)a) | 71.8±13.3 | 73.1±13.7 | 71.1±12.8 | NS |
| Gender (female) | 280 (75) | 95 (72) | 185 (76) | NS |
| Past fracture history | 72 (19) | 23 (17) | 49 (20) | NS |
| Glucocorticoid use | 48 (13) | 15 (11) | 33 (14) | NS |
| Rheumatoid arthritis | 25 (7) | 7 (5) | 18 (7) | NS |
| Concomitant use of vitamin D or active vitamin D3 | 52 (14) | 13 (10) | 39 (16) | NS |
| Year of initiation | ||||
| 2013 | 4 (1) | 3 (2) | 1 (0.4) | |
| 2014 | 42 (11) | 28 (21) | 14 (6) | |
| 2015 | 14 (4) | 8 (6) | 6 (2) | |
| 2016 | 27 (7) | 17 (13) | 10 (4) | |
| 2017 | 68 (18) | 38 (29) | 30 (12) | |
| 2018 | 91 (24) | 31 (23) | 60 (25) | |
| 2019 | 42 (11) | 8 (6) | 34 (14) | |
| 2020 | 57 (12) | 7 (5) | 50 (20) | |
| 2021 | 31 (8) | 4 (3) | 27 (11) | |
| Follow-up period (mo)b) | 25.7±18.9 | 16.8±14.1 | 30.5±19.4 | <0.01 |
| Postponed casesc) | 46 (12) | 10 (8) | 36 (15) | 0.042 |
| Postponed term | ||||
| 1 mo | 17 (37) | 6 (70) | 11 (29) | NS |
| 2 mo | 16 (35) | 4 (30) | 12 (31) | NS |
| ≥3 mo | 13 (28) | 0 | 13 (36) | 0.024 |
Table 2
Details of non-persistence cases in different age groups (n=154)
| Case of non-persistence | Total | Age (yr) | ||
|---|---|---|---|---|
| ≤59 | 60–79 | ≥80 | ||
| Transfer to another hospital | 49 (32) | 6 | 23 | 20 |
| Change to another drug | 47 (31) | 12 | 21 | 14 |
| Interruption of outpatient visitsa) | 34 (22) | 1 | 12 | 21 |
| Interruption due to dental treatment | 12 (8) | 3 | 3 | 6 |
| Adverse event | 4 (3) | 0 | 2 | 2 |
| Patient’s request | 8 (5) | 2 | 3 | 3 |
| Total | 154 | 24 | 64 | 66 |
Table 3
Reasons for postponement of administration in the non-pandemic and pandemic groups (n=46)
Table 4
Details of non-persistence cases (n=154) in the pandemic and non-pandemic periods
| Non-persistence | Non-pandemic period (January 2013–Februay 2020) | Pandemic period (March 2020–December 2022) |
|---|---|---|
| No. of cases | 132 | 22 |
| Incidence (per year) | 16.1 | 12.9 |
Table 5
Characteristics of postponed and non-postponed cases in the pandemic group
| Characteristic | Postponed (n=36) | Non-postponed (n=208) | p-value |
|---|---|---|---|
| Age (yr) | 67.7±13.8 | 70.7±12.6 | NS |
| Gender (female) | 23 (64) | 162 (79) | NS |
| Follow-up period (mo)a) | 35.7±13.5 | 29.6±20.1 | 0.025 |
References
1. Lane NE. Epidemiology, etiology, and diagnosis of osteoporosis. Am J Obstet Gynecol 2006;194(2 Suppl): S3–11.


2. Jackson RD, Donepudi S, Mysiw WJ. Epidemiology of fracture risk in the Women’s Health Initiative. Curr Osteoporos Rep 2008;6:155–61.



3. Jackson RD, Mysiw WJ. Insights into the epidemiology of postmenopausal osteoporosis: the Women’s Health Initiative. Semin Reprod Med 2014;32:454–62.


4. Svedbom A, Hernlund E, Ivergard M, et al. Osteoporosis in the European Union: a compendium of country-specific reports. Arch Osteoporos 2013;8:137.




5. Penning-van Beest FJ, Erkens JA, Olson M, Herings RM. Loss of treatment benefit due to low compliance with bisphosphonate therapy. Osteoporos Int 2008;19:511–7.




6. Chandran M, Lau TC, Gagnon-Arpin I, et al. The health and economic burden of osteoporotic fractures in Singapore and the potential impact of increasing treatment rates through more pharmacological options. Arch Osteoporos 2019;14:114.



7. Chandran M, Mitchell PJ, Amphansap T, et al. Development of the Asia Pacific Consortium on Osteoporosis (APCO) Framework: clinical standards of care for the screening, diagnosis, and management of osteoporosis in the Asia-Pacific region. Osteoporos Int 2021;32:1249–75.




8. Bekker PJ, Holloway DL, Rasmussen AS, et al. A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. J Bone Miner Res 2004;19:1059–66.


9. Cummings SR, San Martin J, McClung MR, et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med 2009;361:756–65.


10. Lyu H, Zhao SS, Yoshida K, et al. Delayed denosumab injections and bone mineral density response: an electronic health record-based study. J Clin Endocrinol Metab 2020;105:1435–44.




11. Tsourdi E, Zillikens MC, Meier C, et al. Fracture risk and management of discontinuation of denosumab therapy: a systematic review and position statement by ECTS. J Clin Endocrinol Metab 2021;106:264–81.


12. Anastasilakis AD, Evangelatos G, Makras P, Iliopoulos A. Rebound-associated vertebral fractures may occur in sequential time points following denosumab discontinuation: need for prompt treatment re-initiation. Bone Rep 2020;12:100267.



13. Koller G, Goetz V, Vandermeer B, et al. Persistence and adherence to parenteral osteoporosis therapies: a systematic review. Osteoporos Int 2020;31:2093–102.



14. Fahrleitner-Pammer A, Papaioannou N, Gielen E, et al. Factors associated with high 24-month persistence with denosumab: results of a real-world, non-interventional study of women with postmenopausal osteoporosis in Germany, Austria, Greece, and Belgium. Arch Osteoporos 2017;12:58.




15. McClung MR, Wagman RB, Miller PD, Wang A, Lewiecki EM. Observations following discontinuation of long-term denosumab therapy. Osteoporos Int 2017;28:1723–32.




16. Tsourdi E, Langdahl B, Cohen-Solal M, et al. Discontinuation of Denosumab therapy for osteoporosis: a systematic review and position statement by ECTS. Bone 2017;105:11–7.


17. Cramer JA, Roy A, Burrell A, et al. Medication compliance and persistence: terminology and definitions. Value Health 2008;11:44–7.


18. Migliaccio S, Francomano D, Romagnoli E, et al. Persistence with denosumab therapy in women affected by osteoporosis with fragility fractures: a multicenter observational real practice study in Italy. J Endocrinol Invest 2017;40:1321–6.



19. Brown JP, Prince RL, Deal C, et al. Comparison of the effect of denosumab and alendronate on BMD and biochemical markers of bone turnover in postmenopausal women with low bone mass: a randomized, blinded, phase 3 trial. J Bone Miner Res 2009;24:153–61.

20. Silverman SL, Siris E, Kendler DL, et al. Persistence at 12 months with denosumab in postmenopausal women with osteoporosis: interim results from a prospective observational study. Osteoporos Int 2015;26:361–72.




21. Karlsson L, Lundkvist J, Psachoulia E, Intorcia M, Strom O. Persistence with denosumab and persistence with oral bisphosphonates for the treatment of postmenopausal osteoporosis: a retrospective, observational study, and a meta-analysis. Osteoporos Int 2015;26:2401–11.




22. Huang CF, Shiao MS, Mao TY. Retrospective analysis of the effects of non-compliance with denosumab on changes in bone mineral density during the COVID-19 pandemic. Patient Prefer Adherence 2021;15:1579–84.




23. Peeters JJ, van den Berg P, van den Bergh JP, et al. Osteoporosis care during the COVID-19 pandemic in the Netherlands: a national survey. Arch Osteoporos 2021;16:11.




24. Fuggle NR, Singer A, Gill C, et al. How has COVID-19 affected the treatment of osteoporosis?: an IOF-NOF-ESCEO global survey. Osteoporos Int 2021;32:611–7.




25. Guidance for clinicians in management of patients with osteoporosis in the COVID-19 era [Internet] Tokyo: Japan Osteoporosis Society. 2020 [cited 2022 Dec 2]. Available from: http://www.josteo.com/ja/news/doc/200518_2.pdf
26. Nakayama M, Furuya T, Inoue E, et al. Adherence to denosumab for the treatment of osteoporosis in Japanese patients with rheumatoid arthritis: results from the IORRA cohort study. Mod Rheumatol 2021;31:510–3.


27. Kobayashi K, Ando K, Machino M, et al. Persistence of denosumab therapy among patients with osteoporosis. Asian Spine J 2020;14:453–8.




28. Chandran M, Hao Y, Kwee AK, Cheen MH, Chin YA, Ng VY. Adherence to dosing schedule of denosumab therapy for osteoporosis during COVID-19 lockdown: an electronic medical record and pharmacy claims database study from Asia. Osteoporos Int 2022;33:251–61.




- TOOLS