 |
 |
- Search
| Asian Spine J > Volume 17(3); 2023 > Article |
|
Abstract
Study Design
A descriptive cross-sectional study of the anatomical variations, morphometry, and histology of the iliolumbar veins (ILVs).
Purpose
This study aimed to describe the anatomical variations of the ILVs and determine their tissue composition in South African cadavers of European descent.
Overview of Literature
A safe anterior surgical approach to the L4/L5 intervertebral disc space requires understanding the anatomy of the ILVs. Limited understanding of ILVs and their variations may lead to inadvertent avulsion of veins with subsequent hemorrhage and damage to the adjacent nerves intraoperatively. Variations in ILVs are population specific, but such reports are limited in the South African population.
Methods
Eighty-nine adult cadavers were dissected to reveal ILV patterns. The variations (origin, course, and drainage pattern), morphometries, and topography of the ILVs were studied. A total of 19 (10 proximal, nine distal) ILVs were processed for hematoxylin and eosin, Masson’s trichrome, and Verhoeff’s histological staining to determine the tissue composition.
Results
The ILVs were identified in 100% of the cases, and 45% of the ILVs were anastomosed to each other bilaterally. The right-side ILVs terminated into the posterior surfaces of the iliac vessels (p=0.001), whereas the left-side ILVs terminated into the lateral surfaces (p=0.001). The left-side proximal ILVs had higher elastic fiber composition (p=0.030). The ratio of the ILVs’ elastic fibers to collagen fibers was 1:9, and 61% of the cadavers exhibited type 1 ILV pattern. Moreover, 42% of the ILVs were at the S1 vertebral level with 31% lying between L4 and L5 spinal nerve roots. The obturator nerve coursed anteriorly to the ILVs in 96% of cases.
Conclusions
The ILV variations described for South Africans present new additional patterns, such as bilateral anastomosis and laterality of the terminal drainage. The ILVs have more collagen fibers than elastic fibers, predisposing them to avulsion during surgical retraction. The identification of all the ILVs is crucial to minimize inadvertent hemorrhage and damage to adjacent structures.
The anterior surgical approach to the lumbosacral spine is preferred during pelvic and spinal surgeries [1–3]. Vascular injuries during anterior exposure are potentially life-threatening [3]. The mechanisms of injury to vascular structures include excessive traction, avulsion, and/or laceration [2]. Most intraoperative vascular injuries are caused by incorrect identification, dissection, and control of vessels during the exposure phase instead of the surgical procedure. Unknown variations of the typical vascular anatomy contribute significantly to these injuries [4]. Unrecognized vascular variations may also add risks of injury to the surrounding neuronal structures [5]. Unexpected anatomical patterns are known to contribute to approximately 25% of all surgical technical errors [6,7].
The iliolumbar veins (ILVs) are generally encountered during the anterior exposure of the lumbosacral junction and the intervertebral disc space between the fourth and fifth lumbar vertebrae (L4/L5) [8] and are vulnerable during surgery, leading to severe hemorrhage [9]. Therefore, initially identifying and ligating the ILVs to allow for safe exposure and reduction of intraoperative bleeding is recommended [8]. Surgical ligation of the ILVs does not affect the venous circulation of the affected regions because of extensive collateral circulation [1]. Morphometric parameters of the ILVs, such as the length of the trunk and distance to nearby structures, are vital in determining the surgical approach, risk of avulsion, and possible bleeding control methods [8–10]. Topographical characteristics of the ILVs such as their landmarks, number of trunks, and drainage patterns are crucial in identifying and locating the ILVs for ligation during the exposure phase [1,9,10].
Reducing damage to the ILVs during surgery is dependent on detailed anatomical knowledge and preoperative imaging [9,11]. Understanding the detailed anatomy of the ILV is difficult because of the high variability of their anatomical presentations and the paucity of studies on comprehensive anatomical description [1,8,9,12,13]. The variability appears to be population specific [11,14], and under certain conditions, such as surgical operations, a variation may be clinically significant in one population and may not be in another [15]. Knowledge of anatomical variations across populations is essential to clinicians working with diverse populations. Cadaveric dissections provide the best way of describing the detailed anatomy of ILV variations owing to the wide exposure provided by this method [16]. This study aimed to examine the anatomy of ILVs and their histological composition in South African cadavers of European descent.
A total of 89 adult South African cadavers of European descent were used in this study (ethical waiver no., W-CBP-210401-01). All cadavers that presented with anatomical variations of the inferior vena cava (IVC) and the common iliac vein (CIV) and had visible trauma or previous surgery to the lumbar region were excluded from the study. Five of the total dissected cadavers successfully met the exclusion criteria, leaving 84 (43 men and 41 women) for the analysis. The mean age of the cadavers was 75.11±12.57 years.
To study the ILVs, the cadavers were placed in the supine position. A midline incision was made from the xiphoid process to the pubic symphysis to open the anterior abdominal wall. The peritoneum and its contents were moved upward, and the retroperitoneal space was accessed. The origins of the IVC and the abdominal aorta were identified and cleaned. The abdominal aorta and common iliac arteries were removed to expose the lumbar vertebra and corresponding intervertebral spaces. The ILVs were then identified bilaterally. The number, origin, course, and variant patterns of the ILVs were analyzed and recorded. In addition, the drainage sites of each vein were recorded. The obturator nerve (ON) and lumbosacral trunk (LST) were also identified after removing the psoas major (occasionally with psoas minor) muscle. The relationship of the ILVs to the ON and LST was observed and recorded. The distance between the vessels and nerves was measured using a digital Vernier caliper (accuracy ±0.02 mm, resolution ±0.01 mm, repeatability ±0.01 mm) on both the left and right sides. The length of the ILVs was measured using a string, and the distance was transferred to a ruler. The following parameters were measured: (1) distance from the origin of the IVC to the ostium of the ILVs into the iliac veins; (2) distance from the ostium of the ILVs into the iliac veins to the point of intersection with ON (where there was no intersection, the shortest distance from the ostium of the ILV to the nerve was measured); (3) distance from the ostium of the ILVs into the iliac veins to the point of intersection with LST; and (4) distance from the ostium of the ILV to the point where the ILV receives its first tributary (length of the stem/trunk of the ILV).
A total of 19 ILVs, of which 10 were proximal ILVs (PILVs, five men, five women) and nine were distal ILVs (DILVs; five men, four women), were harvested from both the right and left sides of the randomly selected cadavers and stored in 10% formalin-buffered solution. Cadavers who had known vascular diseases at the time of death and those with evidence of gross venous diseases such as obliteration of iliac veins that extended to the ILVs were excluded.
Tissue samples of approximately 8 mm in length were processed for hematoxylin and eosin (H&E), modified Verhoeff’s, and Masson’s trichrome histological staining. Images of stained sections were captured with a digital camera mounted on a light microscope (ZEISS Axioscope; https://www.zeiss.com/microscopy/en/home.html). Stacks of images were analyzed using the ImageJ software (https://imagej.net/). The distance variables were calibrated in ImageJ software before image analysis. Vessel-wall thickness and general appearance were determined on H&E-stained sections. Area percentages of collagen fibers were evaluated from Masson’s trichrome-stained sections (collagen fibers stain green) and area percentages of elastic fibers (stain black) were evaluated from Verhoeff’s-stained sections.
Data were managed in Microsoft Excel 2010 (Microsoft Corp., Redmond, WA, USA) and analyzed using IBM SPSS Statistics for Windows ver. 23.0 (IBM Corp., Armonk, NY, USA). Histograms and the Shapiro-Wilk test were used to test data normality. The comparisons of mean values between the right and left sides and between men and women were obtained using a two-sample Student t-test, and a significance level of p<0.05 was used for statistical inference.
The ILVs were observed in all (100%) specimens examined. A total of 415 (right=209, left=206) ILV trunks were observed. Five different ILV trunks were observed and named the ascending lumbar vein (ALV), ALV–ILV common trunk (ALV–ILV), PILV, DILV, and accessory ILV (AILV). Of these five ILV trunks, the maximum number per side was four and was more apparent in women (p=0.021). The PILV trunks were observed more frequently in 157 sides (93.5%). The frequency of the three main ILV trunks (46.4%) was higher on the right side, whereas the frequency of the two main ILV trunks (50%) was higher on the left side. The majority of the ILVs (56.6%) drained into the CIVs. Drainage of the ILVs into the internal iliac vein (IIV) was significantly higher on the right side (p=0.001), whereas drainage into the external iliac veins (EIVs) was significantly higher on the left side (p=0.001). In one case (0.25%), the ILV drained into both the EIV and IIV. In another case (0.25%), the ILV drained into the IVC. Most (64.8%) of the ILVs had two tributaries, but cases with 1 (14.4%), 3 (17.2%), 4 (2.6%), 5 (0.8%), and 6 (0.2%) tributaries were observed. The presence of five tributaries was significantly higher on the right side (p=0.005). The tributaries of 77 ILVs (45.8%) anastomosed with each other bilaterally (Fig. 1A–D). A large proportion of the ALVs (43.4%) drained the L5 vertebral region. A greater proportion of the ALV–ILVs (42.6%) drained the iliac fossa, iliacus muscle, psoas major muscle, and L5 vertebral region. The majority of the PILVs (55.1%) drained the combination of iliac fossa, iliacus muscle, psoas major muscle, and L5 vertebral region. Most of the DILVs (67%) and AILVs (90%) drained the iliac fossa and iliacus muscle. The direction of entry of the ILVs into terminal drainage was predominant on the posterior surface on the right side (p=0.001) and predominantly laterally on the left side (p=0.001). The classification of the ILVs patterns followed that of Unruh et al. [13]. Type 1 patterns were the most common (60.71%) (Fig. 2A–C), whereas type 2 was the least common (6.55%).
A higher proportion (42.2%) of all the ILVs was found at the level of the first sacral (S1) vertebra (Table 1). The ON coursed anterior to the majority (96.4%) of the ILVs and inferior in 3.6% of the ILVs. Close to a third (30.8%) of the ILVs are found between the L4 and L5 spinal nerve roots of the LST. The majority of the L4 spinal nerve roots of the LST (L4/LST) (67.4%) were anterior to the ILVs (Fig. 3A, B). The L5 spinal nerve root of the LST (L5/LST) coursed posterior to the ILVs in most cases (91.4%). The LST (combined L4 and L5 spinal nerve roots) coursed posteriorly to the ILVs in most cases (93.5%). Generally, the distance from the origin of the IVC to the ostium of the ILV trunks ranged from 19 to 116 mm. About a third (28%) of the ALVs were found <30 mm from the origin of the IVC, most of which were in women (p=0.048), and on the right side of the body. A total of 30.5% of ALV–ILVs on the right side was found <30 mm from the origin of the IVC. The left-side mean distances from the origin of the IVC to the ostium of the ILV trunks were significantly higher for all the ILV trunks (p=0.003 for the ALVs, p=0.002 for the ALV–ILVs, p=0.001 for the PILVs, DILVs, and AILVs). The overall mean length of all the ILV trunks was 14.09±7.15 mm, which ranged from 2.5 to 53 mm. The mean length of the ALV trunks was significantly higher than that of the DILV trunks (p=0.039). Inferiorly located ILVs lie close to the ON and LST (Table 2). The distance from the ON to the ILV ranged from 0 to 62.7 mm. The distance from the LST to the ILV ranged from 0 to 61.9 mm. The ON intersected more frequently with the inferiorly located ILVs. Most of the LST intersections (89.8%) occurred at the ostium of the ILVs.
The ILVs exhibited all three tunics (Fig. 4A–C). The overall mean wall thickness of the PILVs was 0.22±0.06 mm, and the left-side mean was marginally higher (p=0.051). The overall mean wall thickness of the DILVs was 0.23±0.06 mm. All ILVs had elastic and collagen fibers. The overall mean for the elastic fiber percentage area was 1.38%±0.50% (PILVs 1.33%±0.47%, DILVs 1.43%±0.53%). The left-side PILVs had more elastic fibers than the right-side PILVs (p=0.03). The overall mean for the collagen fiber percentage area was 12.48%±4.26% (PILVs 12.66%±4.35%, DILVs 12.30%±4.18%). The ratio of the overall elastic fibers to the collagen fibers was 1:9.1 (1.38:12.48).
This study examined the anatomical variations and tissue composition of the ILVs in South African cadavers of European descent. This information is vital for preoperative planning, particularly when determining the vessels for ligation during lumbosacral surgery [1,11]. The anatomy and tissue composition of the ILVs in South African populations and other African populations has not been explored before. The observation of ILV trunks in this study concurs with the findings from similar cadaveric studies that have reported 100% occurrence of the ILV trunks in the Turkish, Chinese, and British populations [12,14,17]. However, variable frequencies on the presence of the ILVs have been reported elsewhere, although the total absence of the ILVs is very unlikely [1,9,11,18]. Caution should be taken when interpreting radiological images, as some ILVs may not be detected in preoperative phlebography or computed tomography angiography [19,20]. This study confirms that the maximum number of the ILV trunks that surgeons should anticipate on each side is four [13]. The prevalence of a single ILV trunk in the present study is lower than those reported in previous studies on British, French, and American populations [1,8,18]. Additionally, a single ILV trunk was present bilaterally, and this observation contradicts previous observations from the Indian populations that a single ILV trunk is found only on the right side of the body [21].
Knowledge of the ILV wall thickness may help in determining the vessels that may likely be difficult to control for bleeding in the event of hemorrhage [12]. In the present study, the left PILVs showed high mean wall thickness and contained more elastic fibers than the right PILVs and DILVs. Therefore, inadvertent hemorrhage from the PILVs on the left side may potentially be easier to control than from those on the right side [22]. The varying proportions of both elastic and collagen fibers in veins may indicate that the vessels are prone to avulsion tearing and rupture during the retraction of the great vessels to expose the lumbosacral spine [23]. The ILVs with more collagen fibers than elastic fibers are more prone to avulsion and should be ligated [10].
Similar to previous studies, the present study showed that the ILVs drain into the CIV, EIV, and IIV [1,12,24]. However, the CIV has been reported as the sole terminal drainage site for the ILVs with varying frequencies (Table 3) [8,14,17,18]. Similarly, the drainage of the ILVs into the IIV was more frequent on the right side of the body, whereas the drainage of the ILVs into the EIV is more frequent on the left side [1,12]. In the present study, two unique cases of the ILVs draining into the IVC and both the IIV and EIV were observed, and no previous studies have reported this finding. Although rarely encountered, such presentations may increase the risks of iatrogenic injuries, and the resultant damage may lead to excessive hemorrhage caused by connections to two iliac vessels and the IVC [10,12].
The left side was reported to have more ILV tributaries than the right side in the American and Indian populations [9,21]. On the contrary, the overall proportions of multiple tributaries in the present study were higher on the right side than on the left side. Moreover, the PILVs are the only ILVs that may drain all the structures between L4–S1 vertebral bodies, adjacent iliac fossa, iliacus muscle, and psoas major muscle, either singly or in combination. A small proportion of DILVs (5%) on the left side drained the region of the L5 vertebra body, and this contrasts with previous reports that DILVs never drain this region [13].
In the present study, nearly half of the study population showed anastomosis between the ILVs. The anastomotic patterns were based either on the communication between the tributaries of the two ILV trunks (single anastomosis) or between tributaries of four trunks present (triple anastomosis). These anastomotic patterns existed with variable frequencies, and no previous studies have reported their presence, despite the literature describing ILVs as forming an “extensive venous system” that can be compared with the observed anastomotic communications [1,10,13]. Venous anastomosis provides the collateral circulation in the case in which a vein is ligated during surgery [1,10].
In the present study, the ILVs on the right side enter their respective terminal drainage posteriorly, whereas the ILVs of the left side enter their drainage laterally. The descriptions of how the ILVs enter their terminal drainage vessels have not been mentioned elsewhere. The sides of the body differ because the CIV on the left side courses more medially to the structures it drains on its course to join the IVC, thus exposing its lateral side for the possible entry of the ILVs. However, the CIV on the right-side courses more anteriorly to the structures that it drains, making its posterior surface a possible entry site for the ILVs.
The type 2 pattern as described by Unruh et al. [13] was more associated with either the left side [25] or the right side [1] (Table 4); however, in the present study, we did not observe any side differences. Similarly, Kiray et al. [12] and Unruh et al. [13] did not observe associations between the variant type and side. Nalbandian et al. [18] reported that the type 3 pattern is more prevalent in men than in women, but this observation was not confirmed in the present study. The present results confirmed the previous observations that the ILVs may be located at any level between the L4 vertebral body and the region below the S1 vertebral body, with over a third of them lying at the level of the S1 vertebral body [9,12,14]. In a few individuals, the ILVs were located above the L5 vertebra, indicating variations in the previously described “triangular search area” for locating the ILVs [17]. The location of the majority of the ILVs below the level of L4/L5 intervertebral disc space may be advantageous as most of them may not be directly encountered during the intervention of the L4/L5 and L5/S1 intervertebral disc spaces [10].
The ON was reported to course anterior to the ILVs in 100% of the cases examined [1,8,12,14]. Teli et al. [21] observed that 55% of the ONs course anteriorly to the ILVs and only 45% posteriorly in the Indian population. In the present study, the majority of the ILVs course anteriorly to the ON, and few were located inferior to the ON. The variant frequencies reported in the literature may be attributed to the differences in the dissection approaches, especially when the search area for the ILVs is narrowed. Narrowing the search area may also fail to identify the superiorly located ILVs and expose them to inadvertent rupture during medial retraction of great vessels [13].
The relationship of the ILVs to the anterior rami of the L4 and L5 spinal nerve roots and LST is comprehensively described for the first time. In the present study, the formations of most of the LSTs (union of the anterior rami of the L4 and L5 spinal nerve roots) were below the level of the sacroiliac joint, and most of the ILVs located superior to this level were found in between the anterior rami of the L4 and L5 spinal nerve roots [26]. The LST coursed posteriorly to all the ILVs in the majority of cases, supporting previous reports [12,13]. In addition, cases whereby 100% of the LSTs pass posteriorly to the ILVs have been described elsewhere [1,14]. Similar to the observation on the ON above, the LST was observed inferior to the ILVs in a few cases. During the search for the ILVs for ligation, surgeons should expect to find ILVs either anterior, posterior, or superior to the LSTs. Specifically, the anterior course of the L4 spinal nerve root predisposes the nerve to damage during ILV ligation [10]. The posteriorly placed L5 spinal nerve root may be damaged by clamps used to control hemorrhage in situations where avulsions of ILVs occurred due to inadequate exposure [8,10].
The risk of avulsion is increased when the distance between the ostium of the ILV and the origin of the IVC is <30 mm [9,11,17]. The ALV and ALV–ILV trunks in the present study may be at a higher risk of avulsion because of their closeness to the origin of the IVC [8,9,11,14,17]. This study substantiates reports that the distance from the origin of the IVC to the ostia of the ILVs is significantly longer on the left side than on the right side [1,9,11–13]. Except for the PILVs on the right side, the present study supports that the ILV trunks on the left side are usually longer than those on the right [9,11,13]. The inferiorly located ILVs (DILV and AILV) may have a shorter length, and their ligation may increase the risk of sidewall injuries to the iliac veins leading to profuse hemorrhage [8,10].
The present findings concur with reports that the distances between the ON to the ILV trunks are longer in men than in women, but the differences are not significant [13]. In addition, the range of distances between the ON and the ILV trunks is wider on the left side than on the right side [13]. Thus, the left-side retroperitoneal surgical approach has less risk of iatrogenic injuries to the ON during the exposure of the L4/L5 intervertebral disc spaces [10]. Furthermore, the ON intersects with all the ILVs along its course [8,13,14]. Therefore, it is essential to identify the ON during the ligation of the ILVs to avoid accidental injuries [10,14]. The overall mean distances of the ILVs to the LST observed in the current study correspond to previously reported dimensions [13,14]. The mean distances from the LST to both the ALV and ALV–ILV trunks were significantly higher on the right side, and this observation validated previous reports [13].
Variations in the pattern and topography of the ILVs may be due to aberrant venous embryologic development of the abdomen and pelvic region. The variations may be triggered by genetic, chromosomal, or environmental factors [15]. The CIVs and ILVs are derived from the remnants of the most caudal portion of the posterior cardinal veins [27,28]. The ALV are derived from the supracardinal veins [29]. Any maldevelopment of the supracardinal and posterior cardinal venous anastomosis may lead to variations in the ILV trunks [28]. For instance, anastomosis of the caudal posterior cardinal and caudal supracardinal veins may lead to the development of an ALV–ILV common trunk, whereas failure of the caudal supracardinal and caudal posterior cardinal veins to anastomose may result in ALVs and ILVs having separate trunks [27].
This study shows that the topography of the ILVs is very variable, with new patterns such as bilateral anastomosis of ILVs, ILVs coursing in between the L4 and L5 spinal nerve roots of the LST, and laterality of the terminal drainage. The ILVs have more collagen fibers than elastic fibers in their walls, and this poses a risk of avulsion when retracted intraoperatively. Further studies in distinct populations may be required to validate and expand on the topography and variations of the ILVs to minimize iatrogenic injuries.
Acknowledgments
The authors sincerely thank those who donated their bodies to science so that anatomical research could be performed. Results from such research can potentially increase mankind’s overall knowledge that can improve patient care. Therefore, these donors and their families deserve our highest gratitude. The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Fig. 1
Photographs showing the anastomosis of the iliolumbar vein (ILV). Anastomosing tributaries are pointed with black arrows. (A) No anastomosis. (B) Single anastomosis. (C) Double anastomosis. (D) Triple anastomosis. IVC, inferior vena cava; ALV, ascending lumbar vein; PILV, proximal iliolumbar vein; R.CIV, right common iliac vein; L.CIV, left common iliac vein; ALV-ILV, ascending lumbar vein and iliolumbar vein common trunk; DILV, distal iliolumbar vein; AILV, accessory iliolumbar vein; LEIV, left external iliac vein.
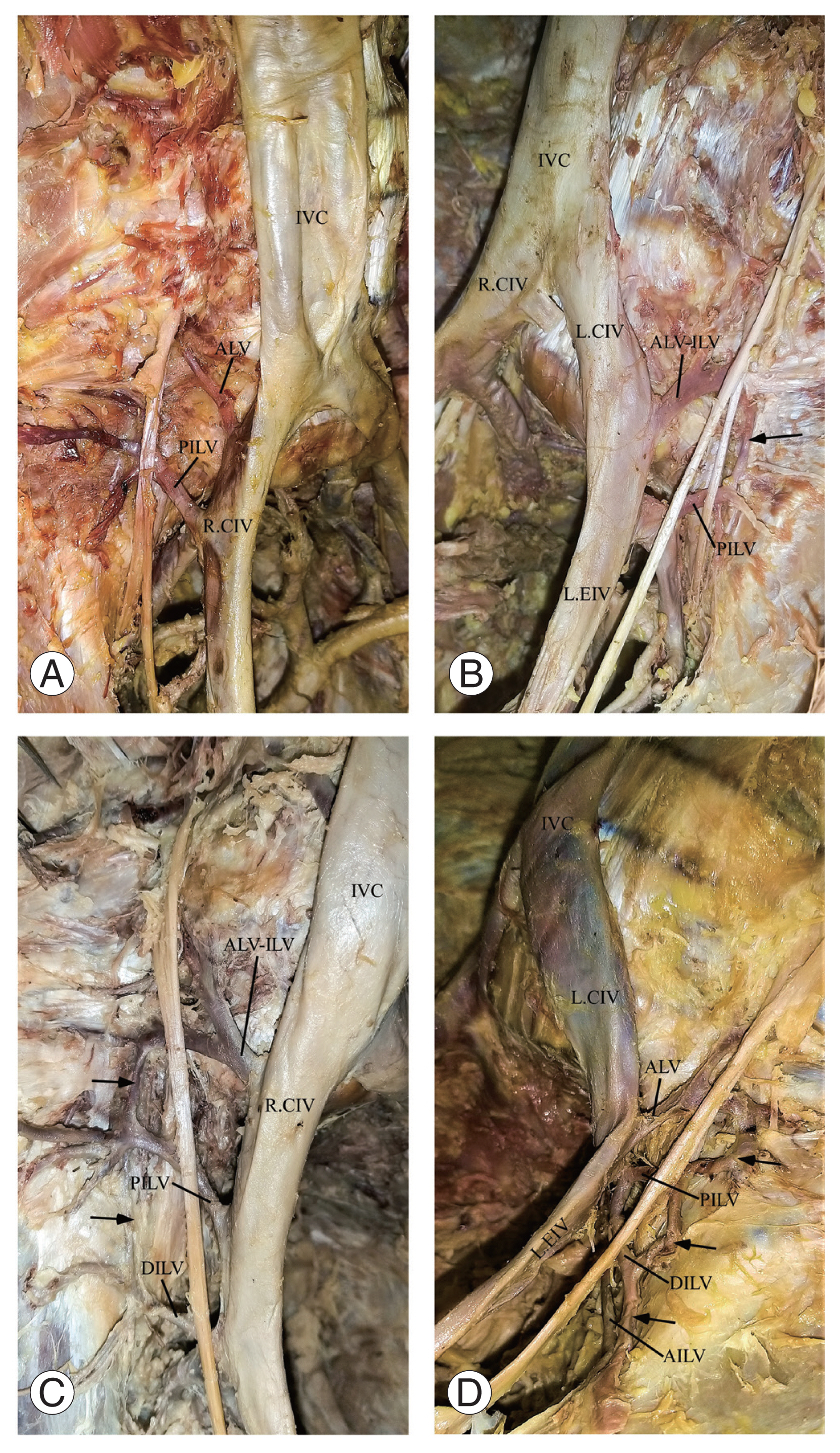
Fig. 2
Photographs showing the classification of iliolumbar veins according to Unruh et al. [13]. (A) Type 1, (B) type 2, and (C) type 3. IVC, inferior vena cava; PILV, proximal iliolumbar vein; DILV, distal iliolumbar vein; R.CIV, right common iliac vein; ALV-ILV, ascending lumbar vein and iliolumbar vein common trunk; L.CIV, left common iliac vein; R.IIV, right internal iliac vein; ALV, ascending lumbar vein.
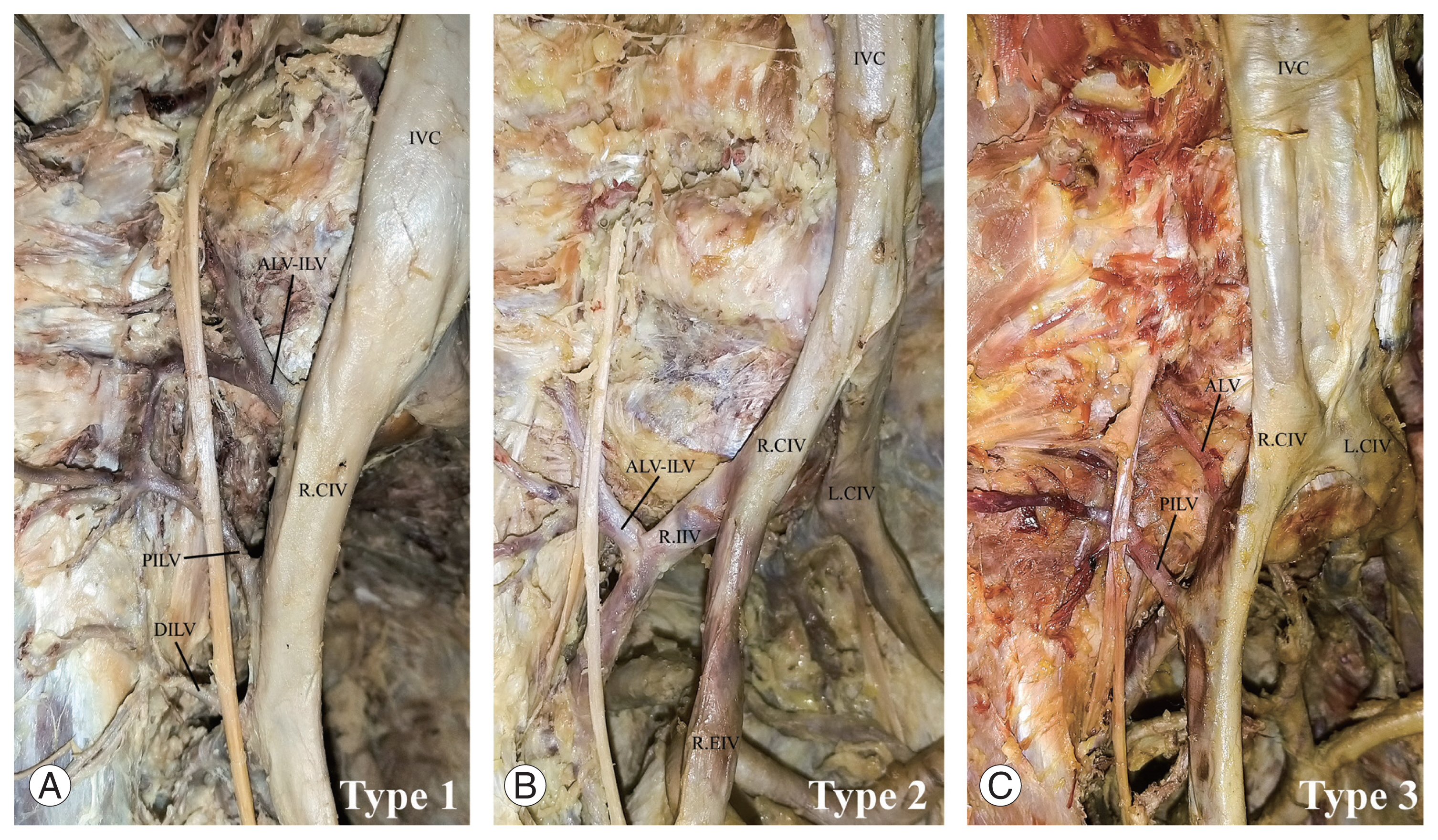
Fig. 3
Photographs showing the relationship between the lumbosacral trunk (LST) and iliolumbar veins (ILV). (A) LST coursing posterior to ILVs and (B) ILV sandwiched between the L4 and L5 spinal nerve roots of the LST. IVC, inferior vena cava; ALV, ascending lumbar vein; PILV, proximal iliolumbar veins; ON, obturator nerve; R.CIV, right common iliac vein; R.EIV, right external iliac vein; L4, fourth lumbar vertebra; L5, fifth lumbar vertebra; L4-LST, L4 spinal nerve root of the lumbosacral trunk; L5-LST, L5 spinal nerve root of the lumbosacral trunk.
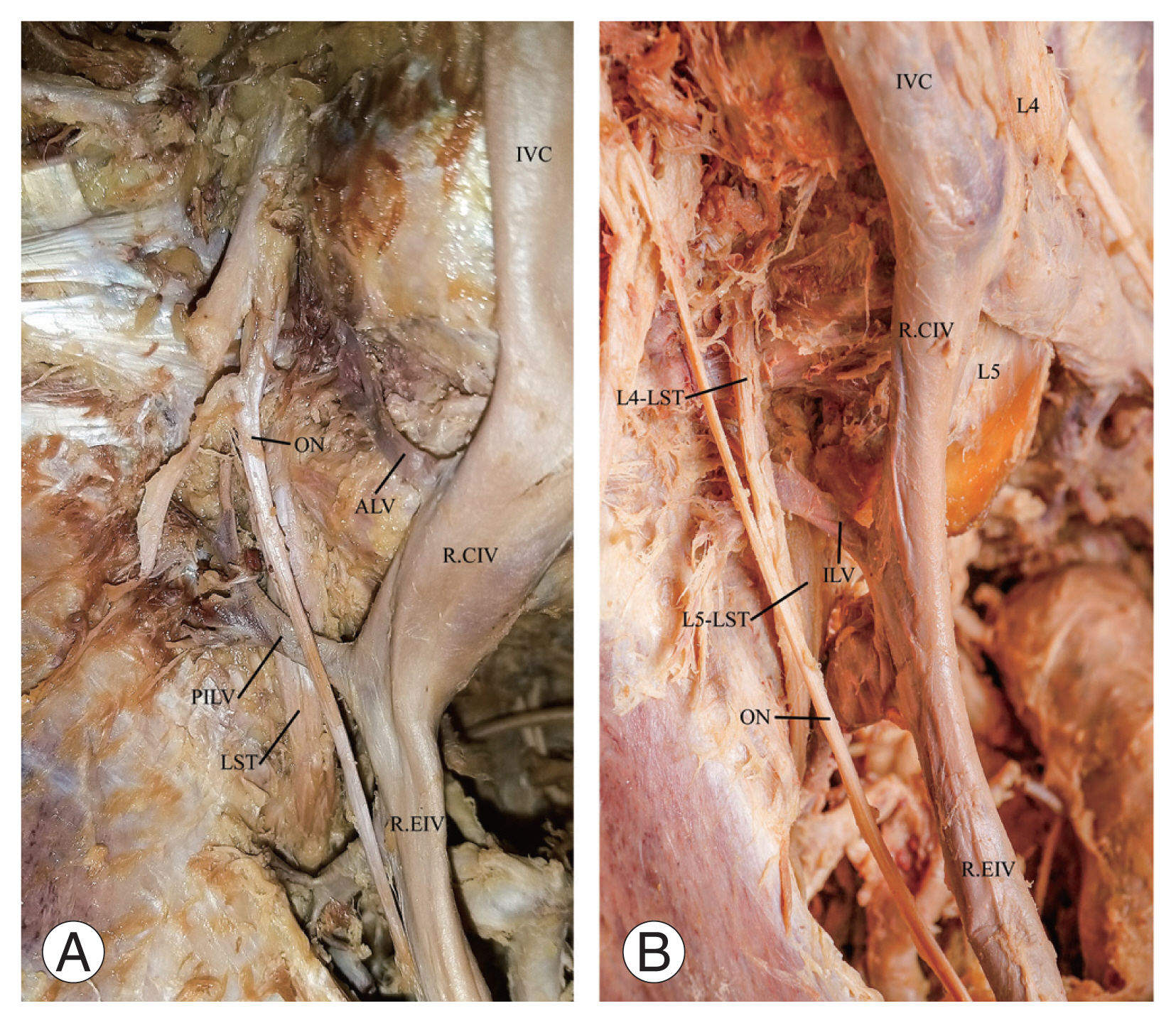
Fig. 4
A photomicrograph of the tissue structure of iliolumbar veins. (A) An H&E-stained section showing the presence of all tunicae in the walls of the iliolumbar veins. (B) A Masson’s trichrome stained section, black dashed arrows showing the presence of collagen fibers (green color) in the layers of iliolumbar veins. (C) A Verhoeff’s-stained section showing the presence of elastic fibers (black color). The red dashed arrows showing scattered elastic fibers. SM, smooth muscle.
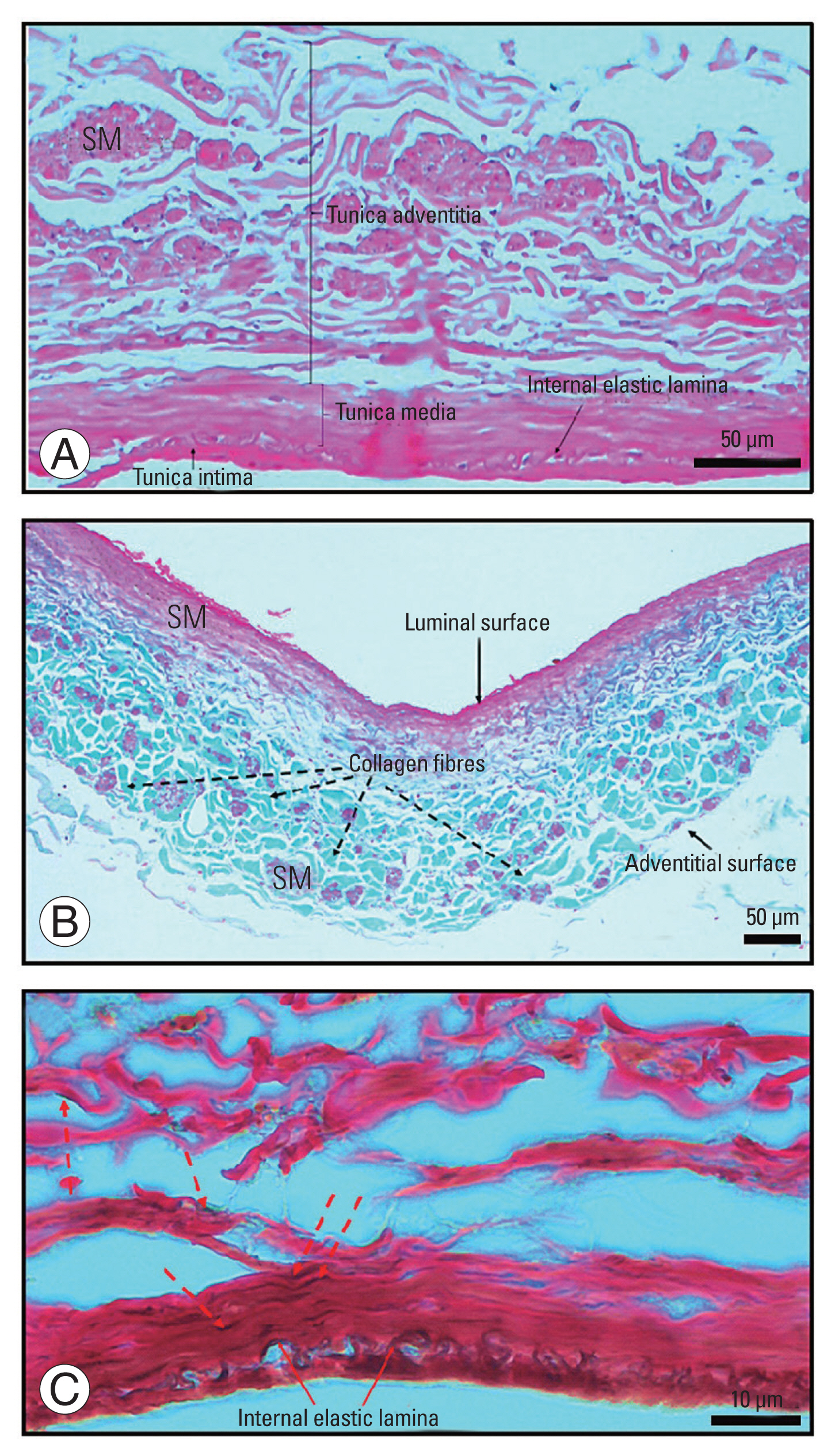
Table 1
The table shows the location of the ILVs with respect to the lumbosacral spine
Table 2
Mean distances from the ostia of ILVs to the obturator nerve and lumbosacral trunk
| ILV trunk | Mean distance and standard deviations (mm) | |||
|---|---|---|---|---|
| Obturator nerve | L4 spinal nerve root of LST | L5 spinal nerve root of LST | Lumbosacral trunk (L4 and L5) | |
| ALV | 22.13±10.95 | 21.72±8.8 | 23.68±12.32 | 27.99±13.58 |
| ALV-ILV | 13.42±6.45 | 15.90±5.57 | 16.85±6.60 | 11.68±6.45 |
| PILV | 5.5 (IQR, 10.1)a) | 10.50±4.07 | 10.75±4.24 | 6.92±6.1 |
| DILV | 2.2 (IQR, 8.6)a) | 6.88±7.4 | 8.38±2.4 | 7.55±5.32 |
| AILV | – | 9.2±0.0 | 11.5±0.0 | 9.94±7.53 |
Table 3
The table shows the terminal drainage of ILVs across the studied populations
| Study (author and year) | CIV (%) | EIV (%) | IIV (%) | IIV & EIV (%) | IVC (%) |
|---|---|---|---|---|---|
| Arthornthurasook and Gaew-Im [25] (1987) | 100.0 | - | - | - | - |
| Jasani and Jaffray [8] (2002) | 100.0 | - | - | - | - |
| Kiray et al. [12] (2004) | 78.9 | 5.3 | 15.8 | - | - |
| Hamid et al. [1] (2007) | 77.0 | 4.0 | 19.0 | - | - |
| Sivakumar et al. [17] (2007) | 100.0 | - | - | - | - |
| Lu et al. [14] (2008) | 100.0 | - | - | - | - |
| Kunakornsawat et al. [24] (2012) | 75.9 | 21.6 | 2.5 | - | - |
| Nalbandian et al. [18] (2013) | 100.0 | - | - | - | - |
| Davis et al. [9] (2019) | 92.9 | 7.1 | - | - | - |
| Current study | 56.6 | 16.9 | 26.0 | 0.25 | 0.25 |
Table 4
The table shows the Unruh et al. [13] classification of ILVs across the studied population groups
| Study (author and year) | Type 1 (%) | Type 2 (%) | Type 3 (%) |
|---|---|---|---|
| Arthornthurasook and Gaew-Im [25] (1987) | 0 | 42.90 | 57.10 |
| Jasani and Jaffray [8] (2002) | 31.25 | 68.75 | 0 |
| Kiray et al. [12] (2004) | 0 | 26.30 | 73.70 |
| Hamid et al. [1] (2007)a) | 0 | 69.00 | 27.00 |
| Sivakumar et al. [17] (2007) | 0 | 13.00 | 87.00 |
| Lu et al. [14] (2008) | 0 | 26.70 | 73.30 |
| Unruh et al. [13] (2008) | 53.00 | 5.00 | 42.00 |
| Nalbandian et al. [18] (2013)a) | 0 | 73.00 | 25.70 |
| Davis et al. [9] (2019)a) | 0 | 46.70 | 46.70 |
| Current study | 60.71 | 6.55 | 32.74 |
References
1. Hamid M, Toussaint PJ, Delmas V, Gillot C, Coutaux A, Plaisant O. Anatomical and radiological evidence for the iliolumbar vein as an inferior lumbar venous system. Clin Anat 2007;20:545–52.


2. Klezl Z, Swamy GN, Vyskocil T, Kryl J, Stulik J. Incidence of vascular complications arising from anterior spinal surgery in the thoraco-lumbar spine. Asian Spine J 2014;8:59–63.



3. Pelletier Y, Lareyre F, Cointat C, Raffort J. Management of vascular complications during anterior lumbar spinal surgery using mini-open retroperitoneal approach. Ann Vasc Surg 2021;74:475–88.


4. Manunga J, Alcala C, Smith J, et al. Technical approach, outcomes, and exposure-related complications in patients undergoing anterior lumbar interbody fusion. J Vasc Surg 2021;73:992–8.


5. Uhl JF, Gillot C, Chahim M. Anatomical variations of the femoral vein. J Vasc Surg 2010;52:714–9.


6. Ellis H. Medico-legal litigation and its links with surgical anatomy. Surgery (Oxford) 2002;20:1–2.

7. Regenbogen SE, Greenberg CC, Studdert DM, Lipsitz SR, Zinner MJ, Gawande AA. Patterns of technical error among surgical malpractice claims: an analysis of strategies to prevent injury to surgical patients. Ann Surg 2007;246:705–11.

8. Jasani V, Jaffray D. The anatomy of the iliolumbar vein: a cadaver study. J Bone Joint Surg Br 2002;84:1046–9.


9. Davis M, Jenkins S, Bordes S, et al. Iliolumbar vein: anatomy and surgical importance during lateral transpsoas and oblique approaches to lumbar spine. World Neurosurg 2019;128:e768–72.


10. Kim DH, Vaccaro AR, Dickman CA, Cho D, Lee SK, Kim I. Surgical anatomy and techniques to the spine. 2nd ed. Philadelphia (PA): Elsevier Health Sciences; 2013.
11. Lolis E, Panagouli E, Venieratos D. Study of the ascending lumbar and iliolumbar veins: surgical anatomy, clinical implications and review of the literature. Ann Anat 2011;193:516–29.


12. Kiray A, Akcali O, Guvencer M, Tetik S, Alici E. Iliolumbar veins have a high frequency of variations. Clin Orthop Relat Res 2004;(425): 252–7.

13. Unruh KP, Camp CL, Zietlow SP, Huddleston PM 3rd. Anatomical variations of the iliolumbar vein with application to the anterior retroperitoneal approach to the lumbar spine: a cadaver study. Clin Anat 2008;21:666–73.


14. Lu S, Xu YQ, Ding ZH, Wang YL, Shi JH, Zhong SZ. Clinical anatomic study of the lower lumbar anterolateral vein: with respect to retroperitoneal endoscopic surgery. Chin J Traumatol 2008;11:110–3.


15. Sanudo JR, Vazquez R, Puerta J. Meaning and clinical interest of the anatomical variations in the 21st century. Eur J Anat 2003;7(Suppl 1): 1–3.
16. Moore KL, Agur AM, Dalley AF. Clinically oriented anatomy. 8th ed. Philadelphia (PA): Wolters Kluwer; 2018.
17. Sivakumar G, Paluzzi A, Freeman B. Avulsion of ascending lumbar and iliolumbar veins in anterior spinal surgery: an anatomical study. Clin Anat 2007;20:553–5.


18. Nalbandian MM, Hoashi JS, Errico TJ. Variations in the iliolumbar vein during the anterior approach for spinal procedures. Spine (Phila Pa 1976) 2013;38:E445–50.


19. Diesinger Y, Charles YP, Bouaka D, Godet J, Steib JP. Preoperative phlebography in anterior L4-L5 disc approach: clinical experience about 63 cases. Orthop Traumatol Surg Res 2012;98:887–93.


20. Barrey C, Ene B, Louis-Tisserand G, Montagna P, Perrin G, Simon E. Vascular anatomy in the lumbar spine investigated by three-dimensional computed tomography angiography: the concept of vascular window. World Neurosurg 2013;79:784–91.


21. Teli CG, Kate NN, Kothandaraman U. Morphometry of the iliolumbar artery and the iliolumbar veins and their correlations with the lumbosacral trunk and the obturator nerve. J Clin Diagn Res 2013;7:422–6.



22. Sidawy AN, Perler BA. Rutherford’s vascular surgery and endovascular therapy. Philadelphia (PA): Elsevier Health Sciences; 2018.
23. Latimer CA, Nelson M, Moore CM, Martin KE. Effect of collagen and elastin content on the burst pressure of human blood vessel seals formed with a bipolar tissue sealing system. J Surg Res 2014;186:73–80.


24. Kunakornsawat S, Prasartritha T, Korbsook P, Vannaprasert N, Tungsiripat R, Tansatit T. Variations of the iliolumbar and ascending lumbar veins. J Spinal Disord Tech 2012;25:433–6.


25. Arthornthurasook A, Gaew-Im K. Study of ascending lumbar and iliolumbar veins. Spine (Phila Pa 1976) 1987;12:70–2.


26. Waikakul S, Chandraphak S, Sangthongsil P. Anatomy of L4 to S3 nerve roots. J Orthop Surg (Hong Kong) 2010;18:352–5.



27. Devi VS. Inderbir Singh’s human embryology. 11th ed. New Delhi: JP Medical Ltd; 2017.
28. Sadler TW. Langman’s medical embryology. 14th ed. Philadelphia (PA): Lippincott Williams & Wilkins; 2018.
29. Maynard RL, Downes N. Anatomy and histology of the laboratory rat in toxicology and biomedical research. London: Academic Press; 2019.
-
METRICS

- Related articles in ASJ
-
Prognostic Factors of Neurological Complications in Spinal Surgeries2018 August;12(4)






