 |
 |
- Search
| Asian Spine J > Volume 16(6); 2022 > Article |
|
Abstract
Purpose
We propose a method for screening for low bone mineral density (BMD) among perimenopausal and postmenopausal women using a self-reported questionnaire.
Overview of Literature
Osteoporosis is a major health problem worldwide. However, it is not cost-effective to evaluate BMD in all patients. Although several tools for predicting osteoporosis have been established, they do not focus much on low BMD prior to the development of osteoporosis.
Methods
We retrospectively reviewed the medical records of 198 women aged 40–70 years who underwent mass screening for osteoporosis at our hospital between 2016 and 2019. The BMD values and the following data were collected: age, body mass index, fracture history, lower back pain, height loss, kyphosis, history of fragility fracture, family history of vertebral or hip fracture, and menopause. The reliability of each data point for the young adult mean <80% was calculated using discriminant analysis. Variables with large weight coefficients were selected and scored. This scoring tool was examined, and a cutoff score for predicting the young adult mean <80% was determined.
Results
Sixty-four participants (32.3%) had a young adult mean <80%. According to the weight coefficients, the following five variables were scored as follows: age ≥60 years 3 points, body mass index <22 kg/m2 3 points, lower back pain 1 point, height loss (cm) 1 point, and menopause 1 point. The area under the receiver operating characteristic curve was 0.738 (95% confidence interval, 0.669–0.807). At cutoff scores of ≥5 and <5, the sensitivity was 82.8%, with specificity of 52.0%.
Osteoporosis is a systemic skeletal disease characterized by low bone mass and microarchitectural deterioration of bone tissue, leading to enhanced bone fragility and increased incidence of fracture [1]. In recent years, the indications of spine surgery have extended to older individuals, and the incidence of postoperative complications associated with bone fragility has been increasing [2]. Osteoporosis is generally diagnosed by hip or lumbar bone mineral density (BMD) measurements using dual X-ray absorptiometry (DXA). Who should be examined using BMD measurements remains under debate because of the cost and availability of DXA. Considering the cost-effectiveness, target screening is better than universal screening. To select patients who would benefit from BMD testing, a simple questionnaire can be useful.
Previous studies reported several tools for predicting the risk of osteoporosis [3,4]. However, they did not focus much on low BMD prior to the development of osteoporosis. Nevertheless, approximately four in five postmenopausal women with new fractures had a BMD of more than −2.5 standard deviations (SDs) of the young adult mean (YAM) value [5]. In Japan, a BMD less than 80% of the YAM value (YAM <80%), which is approximately less than −1.7 to −1.8 SDs of YAM depending on the measurement device [6], is defined as “close examination required” in mass screening for osteoporosis based on the Japanese osteoporosis guidelines [7]. It may be clinically meaningful to predict “close examination required” with simple cost-free tools in order to select patients for whom BMD should be measured.
In the current study, we investigated a method for the mass screening of osteoporosis based on a self-reported questionnaire and measurement of BMD to propose a preliminary self-assessment scoring tool for predicting YAM <80% for perimenopausal and postmenopausal healthy women in Japan.
This study was approved by the institutional review board at Nara City Hospital (approval no., NCH 20–46) and informed consent was obtained in the form of opt-out on the website. We retrospectively reviewed the medical records of 198 consecutive participants who underwent Nara City Osteoporosis Mass Screening in our hospital between 2016 and 2019.
Nara City Osteoporosis Mass Screening for perimenopausal and postmenopausal women was provided in Nara city, Nara prefecture, Japan. The inclusion criteria were women who were residents of Nara city, and were 40, 45, 50, 55, 60, 65, and 70 years of age. These women were screened in 29 hospitals in Nara city, including our institute. The screening consisted of a self-reported questionnaire and measurement of BMD using DXA.
The following data were collected based on the self-reported questionnaire: age, body mass index (BMI), history of fracture, lower back pain, height loss, kyphosis, history of fragility fracture, family history of vertebral or hip fracture, and menopause. Participants filled in their age, height, body weight, and provided yes/no answers for each question in the questionnaire. BMD measurements were performed by DXA technology using Lunar Prodigy equipment (GE Healthcare, Tokyo, Japan) on the total proximal femur and/or lumbar spine. The calculated BMD value was automatically converted to the YAM value, which indicated the percentage of the participants’ BMD compared with that of healthy Japanese adults of the same sex; reference ages were 20–40 years for BMD. Based on the diagnostic criteria for osteoporosis outlined by the Japanese Society for Bone and Mineral Research (JSBMR) [6], the lower YAM value of the total proximal femur and/or lumbar spine was selected as the participant’s YAM value for analysis.
The reliability of each data point for the YAM <80% value was calculated using discriminant analysis. For discriminant analysis, the cutoff points of the participants’ ages and BMIs were determined based on the mean scores for age and BMI to establish a predictive scoring system. Based on a mean age of 59 years and a mean BMI of 22.2 kg/m2, 60 years of age and a BMI of 22 kg/m2 were used as cutoff points. Variables with large weight coefficients were selected for our scoring system. Each selected variable was scored according to the value of the corresponding weight coefficient. For the discriminant analysis, a positive score was a prediction of YAM <80%. Thereafter, the prognostic results obtained with this scoring tool were examined, and a cutoff score was determined. Additionally, the determined cutoff score was examined for the prediction of YAM ≤70%, which is approximately less than −2.5 SDs of YAM. Statistical analyses were performed using IBM SPSS ver. 25.0 for Windows (IBM Corp., Armonk, NY, USA).
A summary of the characteristics of the participants is shown in Table 1. Sixty-four participants (32.3%) had a YAM <80% (representing “close examination required”) and 134 (67.7%) did not. Twenty-eight (14.1%) participants had a YAM ≤70%.
The results of the discriminant analysis (weight coefficients) are presented in Table 2. To obtain these results, five variables with higher values of more than 0.1 (age ≥60 years, BMI <22 kg/m2, lower back pain, height loss, and menopause) were selected to constitute a predictive scoring system. These selected variables were assigned certain integer values according to their weight coefficients. The five variables were scored as follows: age ≥60 years, 3 points; BMI <22 kg/m2, 3 points; lower back pain, 1 point; height loss, 1 point; and menopause, 1 point (Table 3). Discriminant analysis revealed that a higher total score led to a higher expectation of YAM <80% with “close examination required,” with an area under the receiver operating characteristic curve of 0.738 (95% confidence interval, 0.669–0.807) (Fig. 1). A distribution map of the scoring system is shown in Fig. 2. At cutoff scores of ≥5 and <5, the sensitivity was 82.8%, with specificity of 52.0%. For YAM ≤70%, at cutoffs of ≥5 and <5, the sensitivity was 85.7%, with specificity of 54.7%.
In this study, self-reported questionnaire results, including age ≥60 years, BMI <22 kg/m2, lower back pain, height loss, and menopause, provided useful indicators of low BMD (i.e., less than 80% of the YAM value). Our findings suggest that this tool is highly sensitive, convenient, and cost-effective. Of the 81 female participants with scores less than 5, only 11 (13.6%) had a YAM <80%, which was assessed as “close examination required,” and only 4 (4.9%) had a YAM ≤70%, despite the relatively high prevalence of low BMD among all of the participants.
We focused particularly on the answers of participants based on subjective sensations; the analyses were characteristic in the regard that factors other than age were all self-reported. For example, height loss was only based on a subjective assessment rather than a clear-cut criterion such as actual height loss or rate of height loss. Although self-reported height loss is known to be imprecise [8], it is generally impossible to know the true precision of data. In addition, the questionnaire would probably be difficult for most participants to answer if precise data were needed. The same consideration applies to factors other than height loss. In the current study, the scoring system may be useful for analyses, even considering only subjective assessments. Therefore, it could help many women of varied backgrounds to self-assess the risk of loss of BMD.
In the current tool, advanced age, low BMI, lower back pain, height loss, and menopause were all associated with low BMD. Among these factors, lower back pain was the only subjective osteoporotic symptom reported by the patients. Lower back pain is typically reported for osteoporotic vertebral fractures as well as other conditions; for example, patients with osteoporosis occasionally report lower back pain, even if there is no evidence of fracture. Approximately one in 10 patients with osteoporosis without a vertebral fracture are reported to have lower back pain [9]. Bone resorption by activated osteoclasts is thought to lead to such pain [10]. A previous study reported that those with osteoporosis were approximately 2.5 times more likely to experience lower back pain from any causes than patients without osteoporosis [11]; therefore, lower back pain could serve as an indicator of low BMD in this study. Height loss was also included in the scoring system. It is caused by osteoporotic vertebral fractures [8,12,13]. In addition, menopause and advanced age disrupt bone metabolism [14]. Similarly, low BMI disrupts bone metabolism due to decreased fat mass, which contributes to increased bone metabolism via adipocytes [15,16]. Menopause, advanced age, and low BMI are widely known to affect BMD and increase the risk of osteoporosis [17,18]. As mentioned above, all five factors (advanced age, low BMI, lower back pain, height loss, and menopause) were suitable indicators for our tool.
A history of fragility fracture was not included in this tool, although it is one of the JSBMR’s diagnostic criteria for osteoporosis [6], and is adopted in previously established risk tools [4]. Only 14 (7.1%) of the participants had experienced a fragility fracture in our cohort, which was markedly reduced compared with the number of participants with a history of fracture (n=56). It is possible that the participants’ recognition of a fragility fracture was not accurate due to it being self-assessed. Fragility fractures may not be suitable indicators for simple tools based on self-reported questionnaires such as ours, although it may be possible to collect more accurate information about them.
Our findings suggest that the current tool could be useful not only for postmenopausal but also for perimenopausal women. With regard to bone metabolism in women, estrogen obstructs osteoclast activity. However, during the perimenopausal transition, ovarian estrogen production is decreased and osteoclasts are activated, which leads to loss of bone mass [19]. Among perimenopausal women aged 42–52 years, approximately 40% have osteopenia and 10% have osteoporosis [20]. Therefore, perimenopausal women also need to take measures to prevent the loss of bone mass. Although the majority of published tools for assessing the risk of osteoporosis are designed for postmenopausal women, perimenopausal women should be included.
In the current study, the target BMD was 80% of the YAM, which was a higher threshold than that of published risk assessment tools. Identifying patients with a YAM <80% might inform interventions, including treatment and/or close examinations in the early stage of bone loss. For patients whose BMD is lower than normal, treatment should ideally be initiated before BMD falls into the range of osteoporosis, especially considering that most of the structural loss of the bone microarchitecture is not recovered despite treatment [3]. A randomized controlled trial has shown that fragility fractures were significantly reduced in women with osteopenia who had received zoledronate [21]. According to the National Osteoporosis Foundation guidelines published in 2014, women with osteopenia and a high risk of hip fracture should be treated with medication [22]. The current tool improves the identification of patients at an early stage compared with other published tools. In addition, owing to the high thresholds, osteoporosis might rarely be missed. Therefore, we believe that predicting YAM <80% should be clinically meaningful.
In spinal surgery with instrumentation, osteoporosis was reported to be a risk factor for pedicle screw loosening and postoperative vertebral fractures [23–26]. Recognizing low BMD enables not only the enhancement of bone strength by medical treatment before surgery, but also the implementation of measures to increase the pullout strength of pedicle screws during surgery, for example, by using bone cement [27]. Therefore, predicting and detecting low BMD is essential for patients with spine disease being considered for surgery. However, to apply the current preliminary tool to patients with spine disease, further investigation is needed.
There are some limitations to this study. First, as mentioned above, the data may be inaccurate because they are self-reported, which involves potential sources of bias. However, a self-reported questionnaire is an efficient method to collect data from many people quickly and at low cost. In addition, the bias might not be an issue because the objective of this study was to have the participants assess the risk of low BMD. Our tool showed good performance for identifying women at risk of low BMD. Second, the current preliminary scoring system was based on retrospective analyses, and the sample only included women who presented to our hospital, so they may have been particularly health-conscious. These women may thus differ from those in the general population. Further prospective studies with a larger cohort are required to assess the validity of the current study. Finally, we did not evaluate participants’ medical histories related to bone metabolism, such as diabetes or the use of steroids. Patients with such factors should be examined individually.
Our scoring tool using a self-reported questionnaire that included items such as age, BMI, lower back pain, height loss, and menopause performed well for predicting YAM <80% among perimenopausal and postmenopausal women in Japan. This tool may also aid in providing earlier intervention in women with low BMD before they develop osteoporosis. This tool can be a potential convenient and cost-effective screening method for low BMD. Further large-scale research is necessary in future.
Conflict of Interest
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Acknowledgments
The authors thank all members of the outpatient department for orthopedics in our hospital who supported our efforts and helped with this study.
Fig. 1
The receiver operating characteristic curve for the developmental cohort. The area under the curve was 0.738 (95% confidence interval, 0.669–0.807). The straight line represents the diagonal reference line.
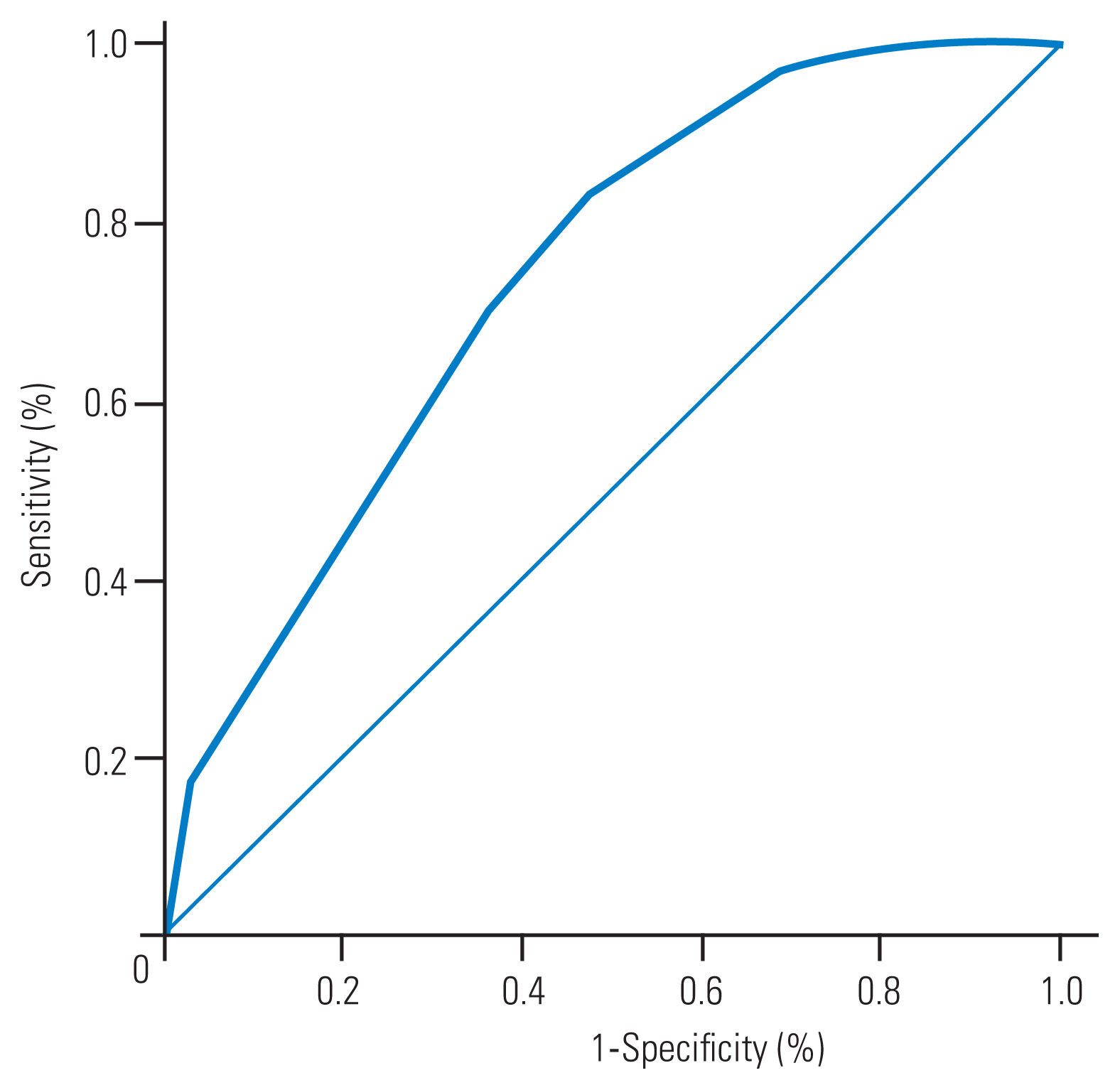
Fig. 2
A distribution map of the scoring system. The total scores of the participants with young adult mean (YAM) ≥80% and YAM <80% are plotted. A total score of less than 5 rarely leads to a YAM<80%.

Table 1
Participants’ characteristics (n=198)
Table 2
Discriminant analysis data (weight coefficient)
References
2. Fujiwara S, Kasagi F, Masunari N, Naito K, Suzuki G, Fukunaga M. Fracture prediction from bone mineral density in Japanese men and women. J Bone Miner Res 2003;18:1547–53.


3. Koh LK, Sedrine WB, Torralba TP, et al. A simple tool to identify Asian women at increased risk of osteoporosis. Osteoporos Int 2001;12:699–705.


4. Sedrine WB, Chevallier T, Zegels B, et al. Development and assessment of the Osteoporosis Index of Risk (OSIRIS) to facilitate selection of women for bone densitometry. Gynecol Endocrinol 2002;16:245–50.


5. Siris ES, Chen YT, Abbott TA, et al. Bone mineral density thresholds for pharmacological intervention to prevent fractures. Arch Intern Med 2004;164:1108–12.


6. Soen S, Fukunaga M, Sugimoto T, et al. Diagnostic criteria for primary osteoporosis: year 2012 revision. J Bone Miner Metab 2013;31:247–57.


7. Committee for Development of Guidelines for Prevention and Treatment of Osteoporosis. Japanese 2015 guidelines for prevention and treatment of osteoporosis. Tokyo: Life Science Publishing; 2015.
8. Briot K, Legrand E, Pouchain D, Monnier S, Roux C. Accuracy of patient-reported height loss and risk factors for height loss among postmenopausal women. CMAJ 2010;182:558–62.



9. Ohtori S, Akazawa T, Murata Y, et al. Risedronate decreases bone resorption and improves low back pain in postmenopausal osteoporosis patients without vertebral fractures. J Clin Neurosci 2010;17:209–13.


10. Fujimoto K, Inage K, Orita S, et al. The nature of osteoporotic low back pain without acute vertebral fracture: a prospective multicenter study on the analgesic effect of monthly minodronic acid hydrate. J Orthop Sci 2017;22:613–7.


11. Chou YC, Shih CC, Lin JG, Chen TL, Liao CC. Low back pain associated with sociodemographic factors, lifestyle and osteoporosis: a population-based study. J Rehabil Med 2013;45:76–80.


12. Kantor SM, Ossa KS, Hoshaw-Woodard SL, Lemeshow S. Height loss and osteoporosis of the hip. J Clin Densitom 2004;7:65–70.


13. Hillier TA, Lui LY, Kado DM, et al. Height loss in older women: risk of hip fracture and mortality independent of vertebral fractures. J Bone Miner Res 2012;27:153–9.


14. Khosla S, Riggs BL. Pathophysiology of age-related bone loss and osteoporosis. Endocrinol Metab Clin North Am 2005;34:1015–30.


15. Fu X, Ma X, Lu H, He W, Wang Z, Zhu S. Associations of fat mass and fat distribution with bone mineral density in pre- and postmenopausal Chinese women. Osteoporos Int 2011;22:113–9.


16. Wu SF, Du XJ. Body mass index may positively correlate with bone mineral density of lumbar vertebra and femoral neck in postmenopausal females. Med Sci Monit 2016;22:145–51.



17. Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause 2010;17:25–54.

18. Fan Z, Li X, Zhang X, Yang Y, Fei Q, Guo A. Comparison of OSTA, FRAX and BMI for predicting postmenopausal osteoporosis in a han population in Beijing: a cross sectional study. Clin Interv Aging 2020;15:1171–80.


19. Armas LA, Recker RR. Pathophysiology of osteoporosis: new mechanistic insights. Endocrinol Metab Clin North Am 2012;41:475–86.

20. Shariati-Sarabi Z, Rezaie HE, Milani N, Rezaie FE, Rezaie AE. Evaluation of bone mineral density in perimenopausal period. Arch Bone Jt Surg 2018;6:57–62.


21. Reid IR, Horne AM, Mihov B, et al. Fracture prevention with zoledronate in older women with osteopenia. N Engl J Med 2018;379:2407–16.


22. Cosman F, de Beur SJ, LeBoff MS, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 2014;25:2359–81.



23. Dvorak MF, Pitzen T, Zhu Q, Gordon JD, Fisher CG, Oxland TR. Anterior cervical plate fixation: a biomechanical study to evaluate the effects of plate design, endplate preparation, and bone mineral density. Spine (Phila Pa 1976) 2005;30:294–301.


24. Reitman CA, Nguyen L, Fogel GR. Biomechanical evaluation of relationship of screw pullout strength, insertional torque, and bone mineral density in the cervical spine. J Spinal Disord Tech 2004;17:306–11.


25. Okuyama K, Abe E, Suzuki T, Tamura Y, Chiba M, Sato K. Influence of bone mineral density on pedicle screw fixation: a study of pedicle screw fixation augmenting posterior lumbar interbody fusion in elderly patients. Spine J 2001;1:402–7.







