Acute Operative Management of Osteoporotic Vertebral Compression Fractures Is Associated with Decreased Morbidity
Article information
Abstract
Study Design
Retrospective national database study design.
Purpose
This study was designed to determine whether acute percutaneous vertebral augmentation (PVA) alters morbidity compared with nonoperative management.
Overview of Literature
Osteoporotic vertebral compression fractures (OCFs) are common and represent a large economic and patient burden. Several recent studies have focused on whether PVA offers benefits compared with nonoperative treatment.
Methods
A retrospective cohort analysis was conducted using the Nationwide Inpatient Sample from 2015 to 2018. Patients with nonelective admissions for OCFs were identified using International Classification of Diseases (10th edition) codes. The exclusion criteria included age of less than 50 years, fusion and decompression procedures, and the presence of neoplasms and infections. Propensity score matching was implemented to construct 2:1 matched cohorts with similar comorbidities at admission. The patients were divided into the operative and nonoperative treatment groups. Univariate and multivariate regression analyses were performed to compare differences in in-hospital complication rates between the groups. All p-values of less than 0.05 were considered significant.
Results
We identified 14,850 patients in the operative group and 29,700 patients in the nonoperative group. In the multivariate analysis, operative treatment was associated with significantly lower rates of pneumonia (odds ratio [OR], 0.75; p<0.001), acute respiratory failure (OR, 0.84; p=0.009), myocardial infarction (OR, 0.20; p<0.001), acute heart failure (OR, 0.80; p=0.001), cardiogenic shock (OR, 0.23; p=0.001), sepsis (OR, 0.39; p<0.001), septic shock (OR 0.50; p<0.001), and pressure ulcerations (OR, 0.71; p<0.001). However, operative treatment was associated with a significantly greater risk of acute renal failure (OR, 1.19; p<0.001) than nonoperative treatment.
Conclusions
Patients who undergo acute PVA for OCFs have lower rates of respiratory complications, cardiac complications, sepsis, and pressure ulcerations while having a higher risk of acute renal failure.
Introduction
Osteoporotic compression fractures (OCFs) are the most common osteoporotic fracture among the aging population, comprising 27% of all osteoporotic fractures [1]. They represent a large economic burden on the healthcare system. In 2015, OCFs had an incidence of 102.1 per 10,000 Medicare beneficiaries, costing approximately $658 million [2]. Of these patients, 35% required acute hospitalization within 7 days following OCF, and 61% required hospitalization greater than 8 days after OCF. Furthermore, the 1-year mortality rate following OCF among Medicare beneficiaries was 21%.
Recent literature has focused on treating OCFs. Operative management consists of percutaneous vertebral augmentation (PVA), including vertebroplasty and kyphoplasty, whereas nonoperative management consists of pain management and external orthoses [3]. Several randomized controlled trials did not show a significant clinical benefit regarding pain or functional outcomes with vertebroplasty [4–7]. In contrast, several studies have found that in the aged population, vertebroplasty offers improved functional outcomes and morbidity and mortality benefits [8,9]. It is not currently recommended in national clinical guidelines for treating OCFs.
Several studies have been conducted to elucidate the benefits of kyphoplasty versus nonoperative treatment for OCFs. Numerous studies have shown that kyphoplasty offers greater improvement in daily activities, quality of life, and vertebral deformity than conservative treatment [10,11]. Patients treated operatively have also been found to have improved mortality benefit [12]. Currently, clinical practice guidelines weakly recommend kyphoplasty [13].
However, the number of studies examining and comparing acute complication rates between operative and nonoperative management of these fractures in patients hospitalized for OCFs is limited. This study was designed to determine whether acute PVA alters morbidity compared with nonoperative management.
Materials and Methods
1. Data source
The Nationwide Inpatient Sample (NIS) is a national database within the Healthcare Cost and Utilization Project published annually and provides nationally representative information on over 7 million hospitalizations across the United States. The NIS is the only national database that uses discharge weights, allowing accurate estimations of nationwide incidence of diseases and procedures. Additionally, the NIS captures all patients, regardless of insurance status. The database uses International Classification of Diseases, 10th revision (ICD-10) codes for documenting all diagnoses and indicated procedures per hospital admission. Given the nonspecific nature of ICD-9 coding used in years before 2015, this study only analyzed NIS years 2015–2018 to use the additional granularity offered by ICD-10 coding. This study did not require Institutional Review Board approval as all patient data within the NIS is deidentified.
2. Patient selection
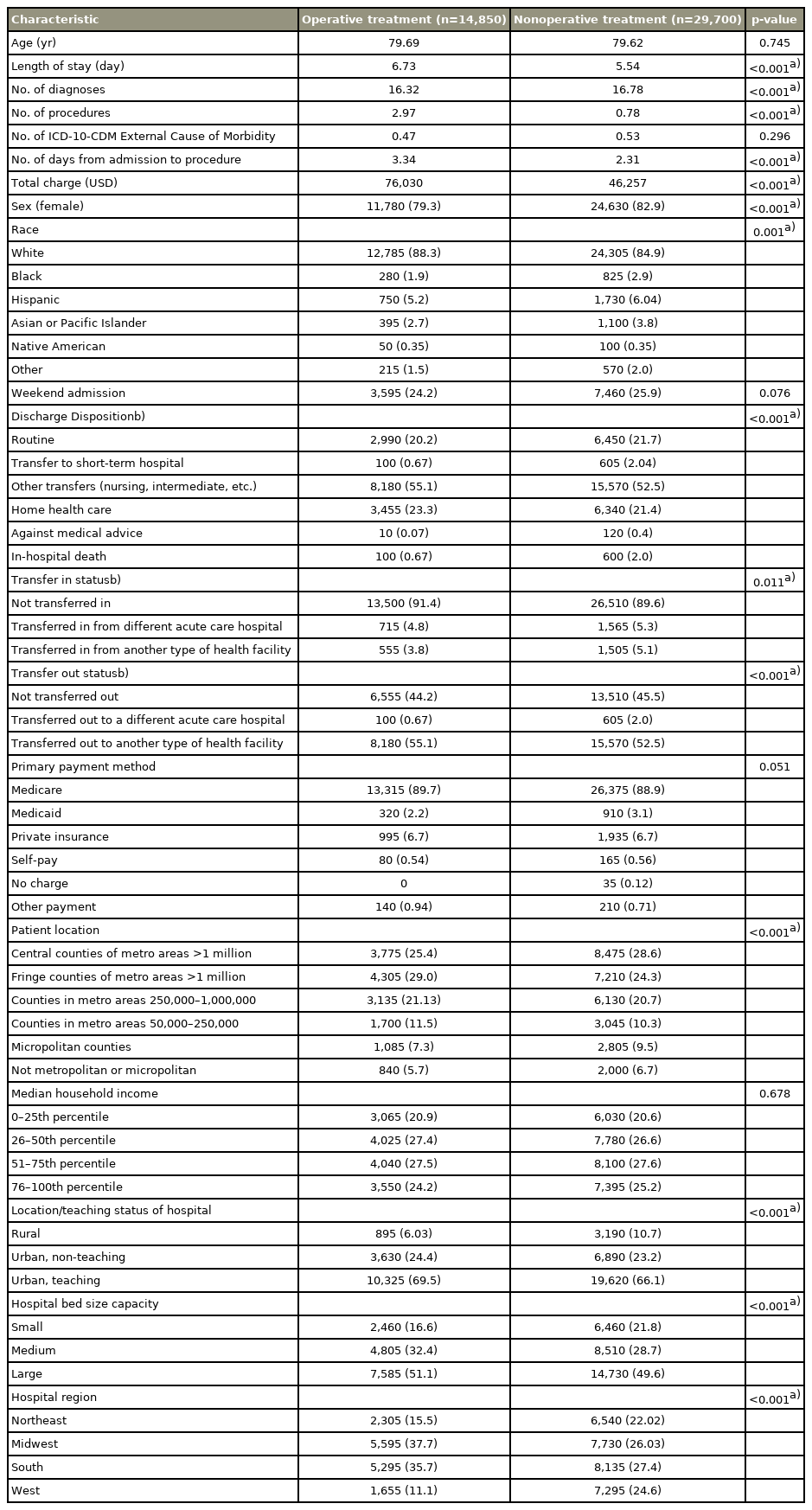
Patients who were nonelectively admitted for OCFs from the 2015 to 2018 NIS dataset were identified using ICD-10 codes. The patients were then divided into those who underwent operative treatment and those who received nonoperative treatment. Operative treatment was defined by the use of PVA, by either vertebroplasty or kyphoplasty. Patients aged less than 50 years, those who were surgically indicated for fusion and decompression procedures, and those with neoplasms and infections were excluded from the study. ICD-10 codes used in defining the diagnosis of osteoporotic compression fracture, the operative treatment group, and exclusion criteria are specified in Table 1.
3. Statistical analysis
Nearest-neighbor propensity score matching 2:1 cohorts for age, total comorbidities, year of procedure, calculated risk of mortality at admission, and calculated illness severity at admission was performed. Univariate analysis was used to assess differences in demographic characteristics, costs, length of stay (LOS), and complication rates between the operative and nonoperative groups. Multivariate logistic regression controlling for significant comorbidities and demographic characteristics was performed to analyze the relationship between either operative or nonoperative OCF treatment and in-hospital complication rates. Significant comorbidities included deficiency anemias, diabetes status, complicated hypertension, fluid and electrolyte disorders, neurological disorders, obesity, renal failure, weight loss, and osteoporosis (Table 2). Significant demographic features included sex, race, in-hospital death, patient location, location/teaching status of hospital, hospital bed capacity, and hospital region (Table 3). All statistical tests were performed using Stata ver. 13.0 (Stata Corp., College Station, TX, USA), and two-sided p-values of less than were used to denote statistical significance.
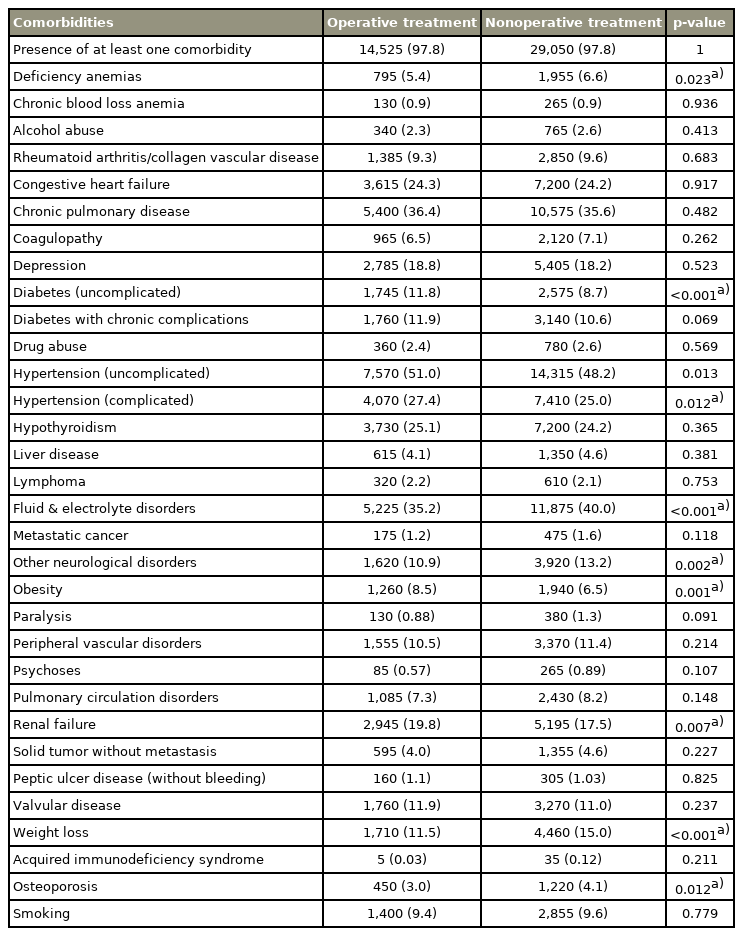
Prevalence of preexisting comorbidities at admission for operative and nonoperative treatment groups
Results
Cohort matching resulted in 14,850 patients who underwent operative treatment and 29,700 patients who underwent nonoperative treatment. The mean age of the patients in the operative (79.3% female) and nonoperative (82.9% female) treatment groups was 79.7 years and 79.6 years, respectively. All demographic and hospitalization characteristics are comprehensively outlined in Table 3.
On univariate analysis, the operative group demonstrated significantly lower rates of respiratory complications, such as pulmonary embolism (PE) (odds ratio [OR], 0.68; 95% confidence interval [CI], 0.56–0.83; p<0.001), pneumonia (OR, 0.63; 95% CI, 0.59–0.68; p<0.001), and acute respiratory failure (OR, 0.84; 95% CI, 0.78–0.91; p<0.001). Operative treatment was also associated with a significantly lower risk of cardiac complications, including myocardial infarction (MI) (OR, 0.34; 95% CI, 0.28–0.42; p<0.001), cardiac arrest (OR, 0.52; 95% CI, 0.35–0.78; p=0.001), acute heart failure (OR, 0.82; 95% CI, 0.75–0.89; p<0.001), and cardiogenic shock (OR, 0.40; 95% CI, 0.23–0.70; p<0.001); moreover, lower rates of cerebral infarction (OR, 0.61; 95% CI, 0.49–0.77; p<0.001) were observed among patients undergoing operative treatment. The rates of infectious sequelae, including sepsis (OR, 0.42; 95% CI, 0.37–0.46; p<0.001) and septic shock (OR, 0.58; 95% CI, 0.46–0.73; p<0.001), were also significantly lower in the operative treatment group. Moreover, the occurrence of pressure ulcers (OR, 0.69; 95% CI, 0.61–0.78; p<0.001) and urinary tract infections (UTI) (OR, 0.92; 95% CI, 0.88–0.97; p=0.001) was significantly decreased in the operative group compared with that in the nonoperative group. Conversely, patients undergoing operative treatment were more likely to experience acute renal failure (OR, 1.10; 95% CI, 1.04–1.16; p=0.001) and radiculopathy (OR, 1.25; 95% CI, 1.09–1.43; p=0.002) than those undergoing nonoperative treatment.
On multivariate analysis, operative treatment continued to demonstrate a significantly decreased risk of complications as previously observed. Significantly lower rates of pneumonia (OR, 0.75; 95% CI, 0.67–0.84; p<0.001) and acute respiratory failure (OR, 0.84; 95% CI, 0.74–0.96; p=0.009) persisted in the operative group. The risk of cardiac complications, including MI (OR, 0.20; 95% CI, 0.15–0.26; p<0.001), acute heart failure (OR, 0.80; 95% CI, 0.69–0.91; p=0.001), and cardiogenic shock (OR, 0.23; 95% CI, 0.10–0.53; p=0.001) remained significantly lower in the operative group, as did the rate of cerebral infarction (OR, 0.44; 95% CI, 0.32–0.62; p<0.001). Operative treatment continued to demonstrate a significantly decreased risk of infectious sequelae, specifically sepsis (OR, 0.39; 95% CI, 0.34–0.45; p<0.001) and septic shock (OR, 0.50; 95% CI, 0.37–0.67; p<0.001), compared with nonoperative treatment. The incidence of pressure ulcers (OR, 0.71; 95% CI, 0.60–0.85; p<0.001) remained significantly lower in the operative group as well. The only complication that had a higher rate in the operative group was acute renal failure (OR, 1.19; 95% CI, 1.08–1.31; p<0.001). No significant differences in the incidence of deep vein thrombosis, PE, cardiac arrest, UTI, and radiculopathy were observed between the two groups. All complication rates and corresponding significance values are listed in Table 4.

Comparison of complication rates between operative and nonoperative treatment groups using univariate and multivariate analysis with corresponding odds ratios and p-values
Operative treatment was associated with a higher mean total cost of admission and mean LOS at $76,030 and 6.73 days, respectively, compared with nonoperative treatment at $46,257 and 5.54 days, respectively (both p<0.001). Among patients who underwent operative treatment, 69.5% were admitted to a metropolitan teaching hospital, 24.4% were admitted to a metropolitan nonteaching hospital, and 6.03% were admitted to a rural hospital compared with 66.1%, 23.2%, and 10.7%, respectively, in the nonoperative group (p<0.001). Overall, nonoperative treatment was associated with a significantly greater in-hospital mortality rate at 2% than operative treatment at 0.67% (p<0.001) (Table 3).
Discussion
In this study, we found that patients hospitalized for OCFs who undergo conservative treatment have an increased rate of acute in-hospital complications, including respiratory complications, cardiac complications, sepsis, and pressure ulcerations, compared with those who undergo acute PVA within 1 week. After PVA, patients are encouraged to mobilize and have been shown to have decreased pain [4,14,15]. Mobilization significantly alters the morbidity and mortality rates [16,17]. This may explain the increased rate of acute complications seen in patients who underwent conservative treatment in this study. Contrarily, patients who undergo PVA have a higher rate of acute renal failure. Acute renal failure is a known postoperative complication, explaining the increased risk in patients treated operatively [18–21].
Our findings are supported by several studies that examined the morbidity and mortality benefits of kyphoplasty and vertebroplasty. Eddin et al. [12] have found that both kyphoplasty and vertebroplasty offer improved mortality benefits compared with nonoperative treatment at up to 4 years following OCF in a national database study. A meta-analysis by Yuan et al. [22] has found that vertebroplasty and kyphoplasty improve quality of life, pain, and functional outcomes. Yang et al. [8] have reported that early vertebroplasty in the aged population offered faster and better pain relief and improved functional outcomes with lower complication rates than conservative therapy. Similarly, Lin et al. [9] have found improved morbidity and mortality with acute versus subacute vertebroplasty in the aged population. Clark et al. [14] have recently published a randomized controlled trial, which showed that vertebroplasty is superior to sham vertebroplasty in reducing pain, improving functional outcome scores, and decreasing analgesic use up to 6 months following OCF.
In a prospective study, Hoshino et al. have found that patients treated with kyphoplasty within 2 months of a painful OCF were less likely to have a decrease in activities of daily living [11]. Similarly, Wardlaw et al. [10] have conducted a randomized clinical trial and found that kyphoplasty resulted in significantly greater improvements in quality of life and disability measures and reduction in back pain than nonoperative treatment in patients with acute painful vertebral fractures. These differences, however, diminished at 12 months. Our findings add to this body of literature, which indicates an improved morbidity benefit with PVA compared with nonoperative management.
Conversely, several publications refute these beneficial outcomes. In a randomized controlled trial, Firanescu et al. [6] have found that vertebroplasty offers no improvements in pain and functional outcomes compared with a sham procedure. Kallmes et al. [7] had similar findings in a separate randomized controlled trial with analogous methods. However, these studies assessed pain and functional outcomes and were not powered to detect differences in morbidity between the two treatment groups.
This study has several limitations. It is a national database study, so it lacks granularity on the patient level. Because of this, we could not report on fracture characteristics or radiographic parameters. An inherent selection bias exists when choosing patients for operative versus nonoperative management. A subset of patients selected for nonoperative management are likely too sick for operative treatment, despite having operative indications. While we matched our cohorts based on several comorbidities and controlled for many potential confounding factors, it is possible that we did not exclude this subset of patients. Additionally, although we believe that early mobilization may contribute to the improved morbidity benefit of acute PVA, we did not have data that support this theory because it is not recorded in the NIS database. Finally, the NIS database is an inpatient-only database, so we could not capture outpatient procedures or long-term complications following initial hospitalization.
Conclusions
Patients who undergo acute PVA for OCF have lower rates of respiratory complications, cardiac complications, sepsis, and pressure ulcerations, while having a higher rate of acute renal failure. This adds to the body of literature that supports that PVA offers morbidity and mortality benefits compared with nonoperative management.
Notes
No potential conflict of interest relevant to this article was reported.