Factors Affecting Incomplete L5/S Posterior Lumbar Interbody Fusion, Including Spinopelvic Sagittal Parameters
Article information
Abstract
Study Design
Retrospective observational study.
Purpose
In this study we identify risk factors, including patient demographics, sagittal parameters, and clinical examinations, affecting incomplete L5/S posterior lumbar interbody fusion (PLIF).
Overview of Literature
The lumbosacral spine is considered to have an interbody fusion rate lower than that of the lumbar spine, but few studies have investigated the cause, including investigating the pelvis. We believe that pelvic morphology can affect L5/S interbody fusion of the lumbosacral spine.
Methods
We observed 141 patients (61 men, 80 women; average age, 65.8 years) who had undergone PLIF and checked for the presence of L5/S interbody fusion. We investigated factors such as age, gender, the presence of diffuse idiopathic skeletal hyperostosis (DISH), fusion level, and grade 2 osteotomy, as well as pre-, post-, and post–preoperative L5/S disk height and angle, lumbar lordosis, Visual Analog Scale (VAS) score, Japanese Orthopaedic Association (JOA) score, and pelvic incidence (PI), comparing those with and without L5/S interbody fusion. In addition, we analyzed the patients classified into short-level (n=111) and multi-level fusion groups (n=30).
Results
Overall, the L5/S interbody fusion rate was 70% (short-level, 78%; multi-level, 40%). Age and pre- and post–preoperative L5/S disk angle were significantly different in each fusion level group. DISH presence, grade 2 osteotomy, and postoperative VAS and JOA scores were significantly different in the short-level fusion group, whereas PI was significantly different in the multi-level fusion group.
Conclusions
Incomplete union after L5/S PLIF correlates with advanced age, many fusion levels, and a large value of preoperative and a small value of post–preoperative L5/S disk angles.
Introduction
Because posterior lumbar interbody fusion (PLIF) simultaneously provides extensive spinal canal decompression and circumferential fusion, it has been performed for various diseases for the past several decades. Although many clinical outcomes have been reported, well-organized investigations of multi-level interbody fusion, including that of the lumbosacral region, are limited.
Since the development of the lateral approach for interbody fusion, minimal invasiveness has progressed, and treatment opportunities for elderly people and patients with multi-level intervertebral lesions are expected to advance in the future. In addition, although the use of spinopelvic parameters has permeated this area of surgery, and the influence of sagittal alignment on clinical outcomes has been widely recognized [1], investigation of the relationship with interbody fusion remains limited. The lumbosacral spine in particular is thought to have a lower rate of interbody fusion than that of the lumbar spine, but few studies have investigated its cause, including examining the pelvis. We believe that pelvic incidence (PI), a pelvic morphology index unique to each individual, can affect fusion of the lumbosacral region.
Therefore, in this study we investigate the physical and surgical factors, as well as the sagittal alignment, that can affect interbody fusion of L5/S PLIF, along with clinical outcomes. We also compare and evaluate the differences in short-level versus multi-level fusion.
Materials and Methods
1. Patient population and inclusion and exclusion criteria
We performed a retrospective observational study conducted in compliance with the Declaration of Helsinki. The study was reviewed and approved by the Institutional Review Board for Clinical Research Ethics at Fujita Health University (approval no., HM 19-123).
Among the 177 patients who had undergone PLIF, including L5/S, between January 2013 and December 2018, 141 patients (61 men, 80 women) were included in our study. We excluded the remaining 36 patients who underwent re-operation, combination with proximal posterolateral fusion, S1 distal instrumentation, and grade 3 osteotomy or higher. We defined diffuse idiopathic skeletal hyperostosis (DISH) as continuous anterior ossification of >4 vertebrae, following the criteria of Resnick et al. [2] (Fig. 1). Diagnoses were as follows: 57 patients had lumbar canal stenosis, 29 had degenerative spondylolisthesis, 24 had lumbar disk hernia, 14 had degenerative lumbar scoliosis, 14 had isthmic spondylolisthesis, and three had other diagnoses. Average patient age was 65.8 years (range, 30–86 years), the average number of fusion levels was 2.0 (single-level, 45 patients; two-level, 66; three-level, 23; four-level, 6; five-level, 1), and the average observation period was 38 months (range, 12–72 months).
2. Surgical technique
As a general principle, in these procedures, pedicle screws (PSs) were inserted after cranial laminectomy and partial facetectomy. For disk space purification, the PS-attached device was distracted, followed by disk space packing with autogenous local bone. Hydroxyapatite particles were added to supplement the space only for those patients in whom a few local bones were used. Finally, the PS systems were fixed after cage insertion, and posterolateral bone transplantation was performed with only local bone or in combination with hydroxyapatite particles.
Grade 2 osteotomy was performed after PS insertion, and no pedicle fracture occurred in any patient during surgery (Fig. 2). The cage implant material in all but 2 patients was polyether ether ketone (PEEK), and the lordosis angle of all cages was ≤6°.
3. Interbody fusion, patient demographics, sagittal parameters, and clinical examinations
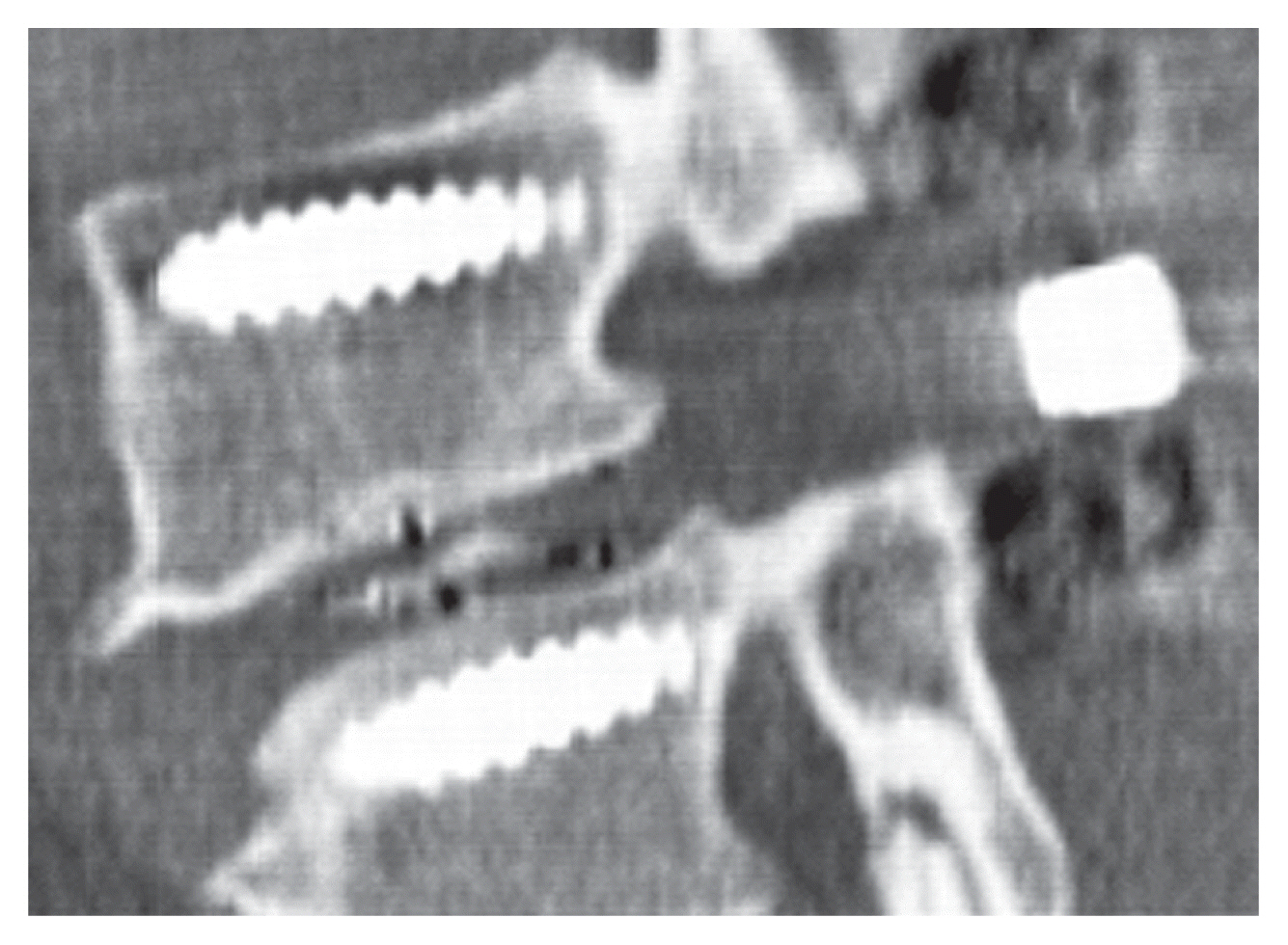
L5/S PLIF was assessed by reconstruction computed tomography (CT) at 1 year postoperatively. In patients for whom fusion was complete (the fusion group), we evaluated the images for the existence of consecutive bone formation in multiple slices obtained around the cage using sagittal and coronal CT. In patients for whom fusion was incomplete (the incomplete-fusion group), we evaluated the images for the existence of a lucent zone or gap around the cage or PS loosening (Fig. 3).

(A) The fusion group was evaluated for the existence of consecutive bone formation with no pedicle screw (PS) loosening and (B) the incomplete fusion group was evaluated for the existence of a lucent zone or gap around the cage or PS loosening in multiple sagittal and coronal computed tomography images.
We measured the spinopelvic sagittal parameters on the lateral whole spine radiograph. We used the Sectra Orthopaedic Package IDS 7 system ver. 11.2 (Sectra AB, Linköping, Sweden) to measure the PI. We measured the L5/S disk angle between the inferior line of the L5 vertebrae and the superior line of the S1 vertebrae and the lumbar lordosis (LL) between the superior line of the L1 vertebrae and the superior line of the S1 vertebrae (Fig. 4). We measured L5/S disk height at the center of the vertebrae. We obtained the postoperative Visual Analog Scale (VAS) scores for low-back pain and the Japanese Orthopaedic Association (JOA) scores at 1 year postoperatively. The JOA score for lumbar pain is based on subjective symptoms and objective findings and restriction of activities of daily living and bladder function; the maximum score is 29 points.

The spinopelvic sagittal parameters were measured on the lateral whole spine radiograph. PI, pelvic incidence; PT, pelvic tilt; SS, sacral slope; SVA, sagittal vertical axis.
We evaluated patient demographics and examination results, including age and gender; DISH presence; fusion level; grade 2 osteotomy; pre-, post-, and post–preoperative L5/S disk height and angle; pre-, post-, and post–preoperative LL and PI; and pre-, post-, and post–preoperative VAS and JOA scores. We compared all demographics and examination results between the patients in the fusion and incomplete-fusion groups, both of which we sub-classified into short-level (≤2) and multi-level (≥3) fusion groups.
4. Statistical analysis
All values are expressed as mean±standard deviation. We performed the statistical analyses with respect to the presence of L5/S interbody fusion. We assessed the continuous variables using the Mann-Whitney U-test and the categorical variables with Fisher’s exact test. We set the level of statistical significance as p<0.05.
We analyzed the variables with statistically significant p-values in the univariable analysis using multivariable analysis, as well as those variables classified in the short- and multi-fusion groups that showed a tendency toward statistical significance. We used receiver operating characteristic (ROC) curve analysis for the continuous variables with statistically significant p-values in the multivariable analysis to determine an optimal cut-off point using the Youden index. We performed the statistical analyses using the JMP ver. 14.2.0 software suite (SAS Institute Inc., Cary, NC, USA).
Results
Overall, the L5/S interbody fusion rate was 70% (99/141): single-level, 91% (41/45); two-level, 70% (46/66); three-level, 39% (9/23); four-level, 50% (3/6); and five-level, 0% (0/1).
In the univariate analysis for L5/S interbody fusion, average patient ages were 61.8 years in the fusion group and 74.0 years in the incomplete-fusion group, which were significantly different (p<0.0001). DISH presence showed no significant difference (p=0.07), with 18.2% in the fusion group and 31.0% in the incomplete-fusion group. Average fusion levels were 1.7 in the fusion group and 2.5 in the incomplete-fusion group, which were significantly different (p<0.0001). At 14.1% in the fusion group and 4.8% in the incomplete-fusion group, grade 2 osteotomy showed no significant difference (p=0.08) (Table 1).

Univariable and multivariable analysis of demographic data in patients with and without interbody fusion
Preoperative L5/S disk angles were 4.9° in the fusion group and 9.3° in the incomplete-fusion group, which were significantly different (p<0.0001). Postoperative L5/S disk angles were 9.6° in the fusion group and 7.9° in the incomplete-fusion group, which were significantly different (p=0.03). Post–preoperative L5/S disk angles were 4.7° in the fusion group and −0.4° in the incomplete-fusion group, which were significantly different (p<0.0001). Post–preoperative LL was 6.2° in the fusion group and 3.6° in the incomplete-fusion group, which was significantly different (p=0.03), whereas pre- and postoperative LL were not significantly different. PI was 50.0° in the fusion group and 54.5° in the incomplete-fusion group, which was significantly different (p=0.01), but PI–pre- and PI–postoperative LL were not significantly different (Table 2).

Univariable and multivariable analysis of sagittal spino-pelvic parameters in patients with and without interbody fusion
The postoperative VAS scores were 11 in the fusion group and 23 in the incomplete-fusion group, which were significantly different (p=0.0006). Pre- and post–preoperative VAS scores were not significantly different. Postoperative JOA scores were 25.8 in the fusion group and 24.0 in the incomplete-fusion group, which were significantly different (p=0.01). Pre- and post–preoperative JOA scores were not significantly different (Table 3).

Univariable and multivariable analysis of clinical data in patients with and without interbody fusion
In the multivariable analysis for L5/S interbody fusion, age (p=0.01), fusion level (p=0.01), pre- and post–preoperative L5/S disk angles (p<0.0002, p<0.0001, respectively), and postoperative VAS scores (p=0.007) were significantly different (Tables 1–3). The optimal cut-off points determined by ROC analysis were as follows: incomplete L5/S interbody fusion, age=71 years, area under the curve (AUC)=0.76; fusion level=2, AUC=0.72; preoperative L5/S disk angle=7.2°, AUC=0.71; and post–preoperative L5/S disk angle=−0.1°, AUC=0.83.
In the univariate analysis classified by short-level and multi-level fusion, age (p=0.0001, p=0.02), preoperative L5/S disk angle (p=0.0002, p=0.01), and post–preoperative L5/S disk angle (p<0.0001, p=0.02) were significantly different in each fusion level group, respectively. DISH presence (p=0.02) and grade 2 osteotomy (p=0.01) were significantly different in the short-level fusion group, and PI (p=0.003) was significantly different in the multi-level fusion group. Postoperative VAS (p=0.001) and JOA scores (p=0.02) were also significantly different in the short-level fusion group (Table 4).
Discussion
Although spinopelvic parameters have become popular for assessing clinical strategy and its results, the reports about assessing interbody fusion are limited [3]. In extensive fusion such as adult spinal deformity surgery, sagittal alignment is particularly important. Above all, the lumbosacral region plays a key role when considering the formation of LL, and it is useful to evaluate the relationship between spinopelvic parameters and L5/S interbody fusion. The L5/S interbody fusion rate is thought to be lower than that of the lumbar spine, but according to previous reports, it can range from 40% to 96% [4–8].
In addition, many reports have discussed the results of up to two-level fusion, whereas for multi-level fusion, most of the results are combined with proximal posterolateral fusion or with distal iliac fusion. Therefore, the reports that precisely evaluate only interbody fusion of multiple levels, including the lumbosacral region, are limited. In this study, we compared the results of L5/S PLIF for multi-level fusion against those for short-level fusion, finding that L5/S PLIF for multi-level fusion is more susceptible to PI than is that for short-level fusion.
Using CT images to evaluate interbody fusion is superior for the early recognition of bridging trabeculation, and these images can be evaluated in greater detail than can X-ray images [9]. We evaluated fusion with CT 1 year after surgery and determined the presence of fusion based on the existence of consecutive bone formation without a lucent zone or gap around the cage. Furthermore, because PS loosening reduces the interbody fusion rate [10], we decided that cases with loosening did not exhibit fusion. In addition, because cage material can affect the evaluation of fusion images, as metals such as titanium can create artifacts [11], and most cages used for the patients in the present study were made of PEEK, with little influence of artifacts, we could evaluate fusion more exactly. As such, we strictly evaluated fusion and concluded that the fusion rate of L5/S PLIF was not high.
Patient and surgical factors can affect bone union after lumbar fusion surgery. One patient factor is age [6], and similar to previous reports, in our study, age was significantly higher in the incomplete-fusion group. DISH, which often is associated with elderly patients, is a risk factor for re-operation after lumbar interbody fusion [12] and for cage retropulsion after PLIF [13]. In the present study, we found that DISH affected interbody fusion for the lumbosacral region for short-level fusion but not for multi-level fusion. The reason may be that the uneven stress between the continuous anterior column caused by DISH and the localized posterior fixation for PLIF might have negative effects on interbody fusion.
Surgical factors include the number of fusion levels, type of grafted bone, and cage material [4,14–17]. Similar to previous reports, in our study, as the number of fusion levels increased, the L5/S interbody fusion rate decreased. We used local bone for grafting in all cases and added hydroxyapatite particles in some cases, but we did not use allogeneic or autologous iliac bone. Because hydroxyapatite alone does not promote interbody fusion [14], the results of fusion are superior with autologous bone as compared to allogeneic bone [15], and PLIF clinical outcomes between autologous local and iliac bone are similar [16]. Thus, we consider that the effects of our surgical method on interbody fusion are localized.
For cage materials, interbody fusion is more likely to be achieved with titanium rather than with PEEK [17], and an elevated cage angle increases fusion rate [18]. Therefore, our cage selection may have affected our clinical outcomes.
Because the lumbosacral spine has larger ranges of anteroposterior flexion and rotation motion than does the lumbar spine [19,20], it is possible that L5/S intervertebral properties can affect interbody fusion. L5/S is likely to be a risk factor of pseudarthrosis after transforaminal lumbar interbody fusion when the preoperative intervertebral height or angle is large [7,21]. Similarly, the patients in the present study were also unlikely to achieve fusion when the preoperative L5/S disk angle was large, and fusion was likely when the postoperative angle was larger than the preoperative angle. Further, the effects of grade 2 osteotomy, which promotes the formation of LL, was recognized only in the short-level fusion group.
As mentioned above, PI affected L5/S interbody fusion during multi-level fusion in our patients. High PI is a risk factor for pseudarthrosis after L5/S anterior interbody fusion [22] and is linked with pelvic tilt (PT) and sacral slope (SS). Because the stress of a long lever arm is concentrated on the lumbosacral spine in multi-level fusion, when PT or SS increases, sagittal shear force also increases and negatively influences L5/S fusion. Therefore, short-level fusion is thought to be easily affected by L5/S disc morphology and sagittal alignment of the lumbar spine, whereas multi-level fusion is thought to be easily affected by L5/S disc and pelvic morphology.
In previous research, poor interbody fusion has been shown to lead to low JOA and JOA Back Pain Evaluation Questionnaire scores [23]. In addition, alignment and bone fusion are involved in the rate of postoperative low-back pain improvement [24]. Meanwhile, interbody fusion and clinical symptoms are not associated [25], and fusion forms such as in-situ union or collapse union are not associated with clinical symptoms [26].
The results for the patients in our study were similar to those of previous reports in that the postoperative JOA score was significantly lower and the postoperative VAS score was significantly higher in the incomplete-fusion group. Although these results were significantly different in the short-level fusion group, they were not significantly different in the multi-level fusion group.
Our study has certain limitations. Ours is a retrospective observational study conducted in a single institution, and the number of cases of multi-level interbody fusion were limited. We could not evaluate the association between interbody fusion and items such as cage installation position or S1 PS insertion position, including bicortical or monocortical fixation. In addition, we did not evaluate other possible factors affecting interbody fusion, such as medical history and bone mineral density.
Conclusions
Incomplete union after L5/S PLIF is correlated with advanced age, many fusion levels, a large value of the preoperative L5/S disk angle, and a small value of the post–preoperative L5/S disk angle. Therefore, we suggest that interbody fusion following L5/S PLIF may be affected by sagittal alignment of the lumbosacral spine in short-level fusion and by pelvic morphology in multi-level fusion.
Notes
Conflict of Interest
No potential conflict of interest relevant to this article was reported.
Author Contributions
Shinichi Kato: conception and design, analysis of data, drafting of the manuscript, critical revision; Nobuki Terada: administrative support; Osamu Niwa: data acquisition; and Mitsuko Yamada: administrative support.