 |
 |
- Search
| Asian Spine J > Volume 16(3); 2022 > Article |
|
Abstract
Purpose
Overview of Literature
Methods
Results
Notes
Author Contributions
Salvador Fuster: senior author and revision; Jaime Jesús Martínez-Anda: clinical charters revision and article writing; Sergio Antonio Castillo-Rivera: clinical charters revision; Caribay Vargas-Reverón: article writing; and Eduard Tornero: revision, statistics, and corresponding author.
Fig. 1
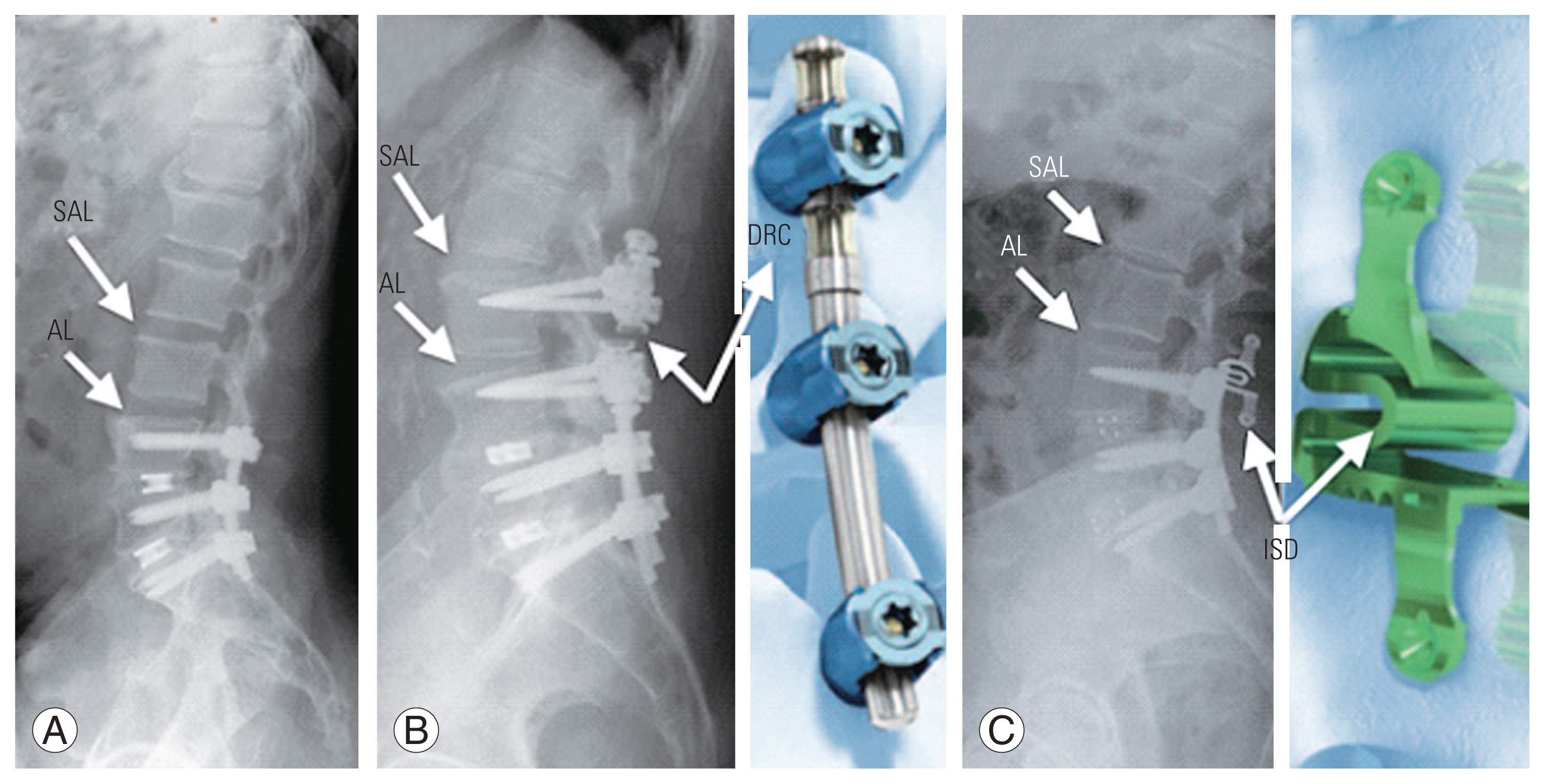
Fig. 2
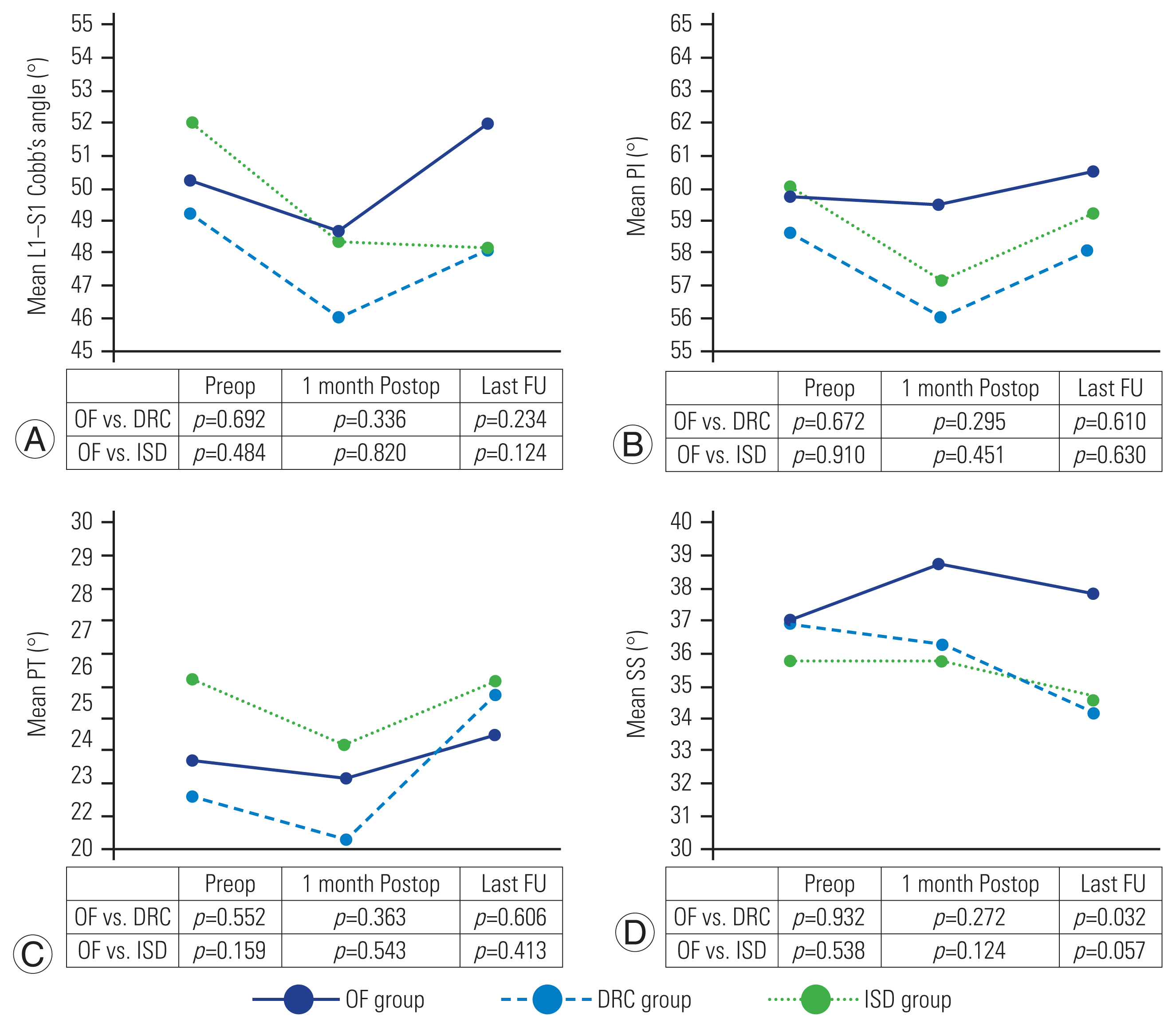
Fig. 3
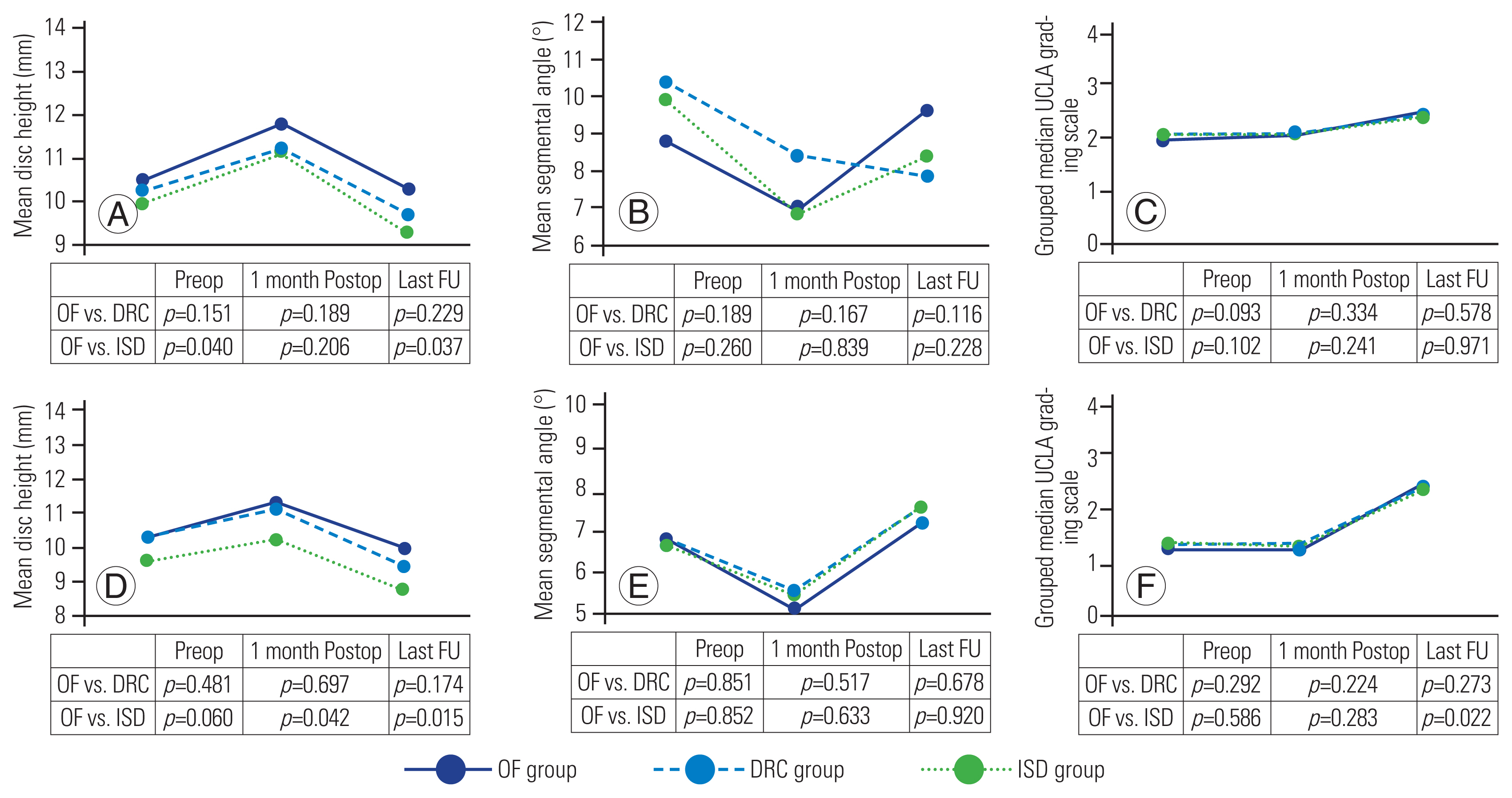
Fig. 4

Fig. 5

Fig. 6

Table 1
| Characteristic | OF group (n=60) | DRC group (n=24) | p-valuea) | ISD group (n=33) | p-valueb) | p-valuec) |
|---|---|---|---|---|---|---|
| Baseline characteristics of patients | ||||||
| Age (yr) | 54.0±13.4 | 59.5±11.0 | 0.078 | 61.7±9.9 | 0.005 | 0.441 |
| Female | 36 (60.0) | 9 (37.5) | 0.062 | 21 (63.6) | 0.730 | 0.051 |
| Body mass index (kg/m2) | 28.0±4.6 | 29.2±2.7 | 0.132 | 25.6±3.7 | 0.013 | <0.001 |
| Smokers | 35 (58.3) | 12 (50.0) | 0.487 | 22 (66.7) | 0.430 | 0.205 |
| Symptoms prior to surgery | 0.590d) | 0.419d) | 0.677d) | |||
| Lumbar pain | 10 (16.7) | 3 (12.5) | 7 (21.2) | |||
| Radicular symptoms | 9 (15.0) | 2 (8.3) | 2 (6.1) | |||
| Both lumbar and radicular | 41 (68.3) | 19 (79.2) | 24 (72.7) | |||
| Duration with symptoms (mo) | 24 (16–40) | 30 (22–46) | 0.292 | 30 (23–36) | 0.170 | 0.980 |
| Type of vertebral fusion | 0.084 | 0.080 | 0.885 | |||
| Single fusion | 35 (58.3) | 9 (37.5) | 13 (39.4) | |||
| Two-level fusion | 25 (41.7) | 15 (62.5) | 0.402e) | 20 (60.6) | 0.016e) | 0.463e) |
| L5–S1 | 13 (21.7) | 3 (12.5) | 5 (15.2) | |||
| L4–L5 | 21 (35.0) | 6 (25.0) | 7 (21.2) | |||
| L3–L4 | 1 (1.7) | 0 | 1 (3.0) | |||
| L4–S1 | 18 (30.0) | 11 (45.8) | 14 (42.4) | |||
| L3–L5 | 6 (10.0) | 2 (8.3) | 0 | |||
| L2–L4 | 1 (1.7) | 2 (8.3) | 6 (18.2) | |||
| Pffirmann preoperative classification >3 at | ||||||
| Index level | 54 (90.0) | 23 (95.8) | 0.667 | 33 (100.0) | 0.086 | 0.421 |
| Adjacent level | 6 (10.0) | 7 (29.2) | 0.043 | 7 (21.2) | 0.210 | 0.491 |
| Supra-adjacent level | 0 | 3 (12.5) | 0.021 | 2 (6.1) | 0.123 | 0.396 |
| Spondylolisthesis prior to surgery | 37 (61.7) | 10 (41.7) | 0.095 | 13 (39.4) | 0.039 | 0.863 |
| Listhesis (%)f) | 18 (10.0–30.0) | 12.5 (8.0–16.5) | 0.102 | 10 (10.0–13.5) | 0.056 | 0.823 |
| Modic changes >1 prior to surgery | 45 (75.0) | 12 (50.0) | 0.027 | 27 (81.8) | 0.045 | 0.011 |
| Outcomes | ||||||
| Follow-up (mo) | 38 (32–45) | 35 (29–36) | 0.002 | 42 (35–71) | 0.010 | <0.001 |
| Bleeding during surgery (mL)g) | 442±327 | 279±144 | 0.049 | 397±256 | 0.644 | 0.164 |
| Duration of surgery (min) | 223±57 | 236±91 | 0.524 | 217±45.3 | 0.607 | 0.360 |
| Fusion achieved | 60 (100.0) | 24 (100.0) | - | 33 (100.0) | - | - |
| ASDe at last follow-up | 38 (63.3) | 16 (66.7) | 0.773 | 26 (78.8) | 0.124 | 0.305 |
| ASDi at last follow-up | 12 (20.0) | 1 (4.2) | 0.097 | 2 (6.1) | 0.127 | 1.000 |
| Required new surgery due to ASDi | 3 (5.0) | 1 (4.2) | 1.000 | 1 (9.1) | 0.931 | 0.618 |
Values are presented as mean±standard deviation, number (%), or median (interquartile range), unless otherwise stated. Statistically significant differences are shown in bold.
OF group, only fusion group; DRC group, dynamic rods construct group; ISD group, interspinous device group; ASDe, adjacent segment degeneration; ASDi, adjacent segment disease (according to criteria defined in methods section).
d) Pearson’s χ2 p-value with 2 degrees of freedom to test the data distribution of all type of symptomatology.






