 |
 |
- Search
| Asian Spine J > Volume 13(2); 2019 > Article |
|
Abstract
Purpose
To compare the clinical and radiological outcomes of patients who underwent anterior cervical discectomy and fusion (ACDF) supplemented with plate fixation using allograft with those who underwent ACDF using tricortical iliac autograft.
Overview of Literature
As plate fixation is becoming popular, it is reported that ACDF using allograft may have similar outcomes compared with ACDF using autograft.
Methods
Forty-one patients who underwent ACDF supplemented with plate fixation were included in this study. We evaluated 24 patients who used cortical ring allograft filled with demineralized bone matrix (DBM) (group A) and 17 patients who used tricortical iliac autograft (group B). In radiological evaluations, fusion rate, subsidence of grafted material, cervical lordosis, fused segmental lordosis, and radiological adjacent segment degeneration (ASD) were observed and analyzed with preoperative and postoperative plain radiographs. Clinical outcomes were evaluated using the Neck Disability Index score, Odom criteria, and Visual Analog Scale score of neck and upper extremity pain. Radiological union was determined by dynamic radiographs using cutoff values of 1 mm of interspinous motion as the indication of pseudarthrosis.
Results
There was no significant difference in the fusion rate, graft subsidence, cervical lordosis, fused segmental lordosis, and ASD incidence between the groups. Operative time was shorter in group A (136 min) than in group B (141 min), but it was not significant (p>0.05). Blood loss was greater in group B (325 mL) than in group A (210 mL, p=0.013). There was no difference in the clinical outcomes before and after surgery.
Since Smith-Robinson [1] first developed anterior cervical discectomy and fusion (ACDF), there have been various modifications and improvements in ACDF, and it is now commonly used as a surgical approach for degenerative cervical spine diseases. The commonly used interbody fusion materials are autograft, allograft, and cage. Among these materials, tricortical iliac autograft has provided the best outcomes with excellent fusion rate, lower proportion of delayed union [2], and good clinical results [3,4]. Recent ACDF studies using allograft have reported excellent clinical outcomes compared with those using autograft [5,6]. Previously, the use of allograft resulted in more cases of nonunion, delayed fusion, graft dislodgement or subsidence, and weakness in mechanical strength compared with the use of autograft. However, recent advances in plate fixation have improved these issues. Furthermore, higher rate of donor site morbidity due to harvesting autograft from anterior iliac crest are frequently reported [7,8], and the longer operative time and increased of bleeding have been considered the drawbacks of ACDF using autograft [9].
Unfortunately, clinical data showing a difference between the use of autograft and demineralized bone matrix (DBM)-filled allograft with plate fixation is limited. Therefore, we compared and analyzed the outcomes obtained using autograft and those using allograft in ACDF with plate fixation.
We reviewed 41 patients who underwent ACDF using plate fixation between April 2010 and August 2013 for treatment of one-level or two-level cervical disc disorder and were regularly followed-up over 24 months (Table 1). Among 41 subjects, 39 patients had cervical spondylotic radiculopathy and two had cervical spondylotic radiculomyelopathy due to a herniated intervertebral disc. Onelevel fusion was performed in 26 patients, whereas 15 patients had two-level fusion. For the operative method, the Smith-Robinson approach was employed [1]. After reaching the anterior portion of the cervical disc, we secured visibility and removed the anterior longitudinal ligament, intervertebral disc, and most of the symptomatic uncinate processes. In group A, 24 patients underwent ACDF with plate fixation using fibula or an ulnar cortical ring allograft filled with DBM (Orthoblast II; IsoTis Orthobiologics, Irvine, CA, USA; DBM with a reverse thermal poloxamer carrier). In group B, 17 patients underwent ACDF with plate fixation using tricortical iliac autograft. The average follow-up period was 39 months (range, 24–59 months). After the operations, patients wore Philadelphia cervical orthoses for 4–6 weeks, and were allowed to do daily cervical motion during their regular visits if they had no serious problems.
The study protocol was approved by the institutional review board of Soonchunhyang University Hospital (IRB approval no., 2017-12-011).
To evaluate clinical outcomes we used the preoperative and postoperative Neck Disability Index (NDI) scores, Odom criteria (Table 2), and Visual Analog Scale (VAS) of neck pain and upper extremity radiating pain. For radiological evaluation, we reviewed the patients’ pre- and postoperative radiographs and collected results of the fusion rate, subsidence of grated material, fused segmental lordosis, and adjacent segment degeneration (ASD).
To evaluate ASD, we compared preoperative radiographs with the final follow-up radiographs, divided into three levels depending on the height reduction of intervertebral disc and whether an osteophyte was formed. If there is no difference between a patient’s preoperative and postoperative radiographs, then the patient is classified as level 1. A patient is classified as level 2 if disc height reduces by ≤50% without posterior osteophyte formation, and as level 3 if disc height reduces by ≥50% with a developed osteophyte. Levels 2 and 3 are identified as showing degenerative change [10]. Radiological union was determined by dynamic radiographs magnified to 150% using cutoff values of 1 mm of interspinous motion as an indicator of pseudarthrosis (Fig. 1).
Subsidence of graft was defined as a change in fused segment height, determined by measuring the distance between the midpoint of proximal vertebral upper endplate and that of lower vertebral lower endplate in fusion segment (Fig. 2). The immediate postoperative measured value was set as the reference value, and we compared the measured data with radiographs taken during each follow-up visit. Cervical lordosis and fused segmental lordosis are measured by Harrison posterior tangent method [11] (Fig. 3).
All statistical analyses were performed using IBM SPSS ver. 21.0 (IBM Corp., Armonk, NY, USA). Ranking data, including NDI and VAS scores, were compared between the groups using Mann–Whitney U-test. Age, operation time, amount of blood loss, subsidence, entire cervical lordosis, and fusion segmental lordosis was compared between groups using paired t-tests pre- and postoperatively to verify the significance of each element. A value of p<0.05 was considered statistically significant.
The VAS scores in the allograft group were 4.4 and 5.7, and those in the autograft group were 4.6 and 5.9 for neck pain and upper extremity pain before the operation, respectively (p=0.844, p=0.820).
During the final follow-up, the neck pain VAS score was not significantly different between the allograft group (1.4) and the autograft group (0.9, p=0.171). Similarly, VAS score for radiating pain was not different between groups (allograft, 0.6; autograft, 1.4; p=0.435). There was no significant difference between groups based on Odom criteria, with 82.6% of the allograft group and 75% of the autograft group scoring good to excellent (p=0.499). The preoperative NDI for the allograft group and autograft group were 26.1 points and 23.5 points, respectively. The postoperative NDI was not significantly different between groups (allograft, 8.9; autograft, 5.0; p=0.262) There was no difference in operation time between groups (allograft, 136 minutes versus autograft, 141 minutes; p=0.734). The allograft procedure showed significantly reduced blood loss compared with autograft (allograft, 210 mL versus autograft, 325 mL; p=0.013).
Mean difference in subsidence of graft was not significantly different between groups (allograft, 4.0 mm; autograft, 2.7 mm; p=0.125) in one-level operations or in twolevel operations (allograft, 1.9 mm; autograft, 2.1 mm; p=0.819). The preoperative fusion segmental lordosis were 5.1° and 4.0° in allograft and autograft (p=0.412) and the postoperative measurements were 5.4° and 5.0° (0.753), respectively. At the final follow-ups, 4.9° and 5.3° were measured for allograft and autograft, with no significant difference (p=0.799).
In terms of entire cervical lordosis, the allograft group showed 11.3° and the autograft group showed 13.5° preoperatively. The cervical lordosis at the final follow-up were 15.0° in autograft group and 17° in allograft group, respectively. The preoperative and postoperative values were not significantly different between groups (p=0.481).
The fusion status of the final follow-up was 94.1% in allograft group, which showed 16 out of 17 patients had achieved union (Figs. 4, 5). In autograft group, 23 out of 24 patients had achieved union with the fusion rate of 95.7%. Fusion status was not significantly different between groups (p=0.663).
For ASD, because the autograft group’s (10/17, 58.8%) follow-up period was longer than that of the allograft group’s, the autograft group (6/24, 25%) demonstrated a higher rate than the allograft. However, when groups were stratified for follow-up period (>2 years or <2 years), there was no significant difference between groups (p=0.387, p=0.202).
Graft incorporation leading to successful spinal fusion requires not only the biological activity of a bone graft but also the condition of the perigraft and its mechanical environment [12]. With its superior biological activity among the known graft materials, tricortical iliac crest autograft was traditionally considered to be the gold standard in ACDF [13]. Additionally, several studies have presented the higher fusion rates using autograft compared with other graft materials [3,14-17]. Although the efficacy of autograft is clearly demonstrated in ACDF, many researchers have noted a growing number of complications using autograft, such as hematoma, fracture, meralgia paresthetica, cosmetic problems, likelihood of increased bleeding, and prolonged operation time [7,8,18,19].
Therefore, recent ACDF studies tend to mainly focus on alternative graft materials with comparable outcomes without the problems related to autograft. As anterior plating becomes more popular, an increasing number of articles support allograft using ACDF with plate augmentation, showing improved postoperative clinical and radiological outcomes [6,18,20-23]. When the plate is augmented, fusion rate was reported to be similar or even better than ACDF without plate fixation [18,24].
Clinically, in our study, a favorable outcome (excellent and good in Odom criteria) was observed in 82.6% of the patients in the allograft group and 75% in the autograft group. This difference is not statistically significant, consistent with the findings of previous studies [22,23].
Previous studies have shown that treatment using cervical plates significantly reduces graft-related complication rates in one- and two-level ACDF with allograft cortical bone, with significantly enhanced arthrodesis [6,21,25]. In particular, Kaiser et al. [21] reported a significant decrease in the graft-related complication rate with anterior plating. Zaveri and Ford [26] emphasized the stabilizing role of the plate which permitted early pain-free mobilization, successfully maintained sagittal cervical spine alignment, and promoted consistent and reliable spinal fusion. Other studies support anterior plating for enhancing the efficacy of autograft or allograft fusions and for reducing the rate of pseudarthrosis and kyphosis [21,27]. However, there are reports of other complications related to plate augmentation, such as dislodgement or breakage of plate, loosening of fixation screws, dysphagia, esophageal injury, and increased rate of delayed union and nonunion caused by stress shielding [26,28]. During the follow-up period, we experienced no notable clinical complications in either group.
In our study, fusion rate was not significantly different between allograft and autograft groups when a plate was augmented in ACDF [22,23,25]. In another study of 540 patients evaluating the efficacy of anterior cervical plate (233 allograft with plate, 289 allograft without plate), fusion rates for one- and two-level ACDF with anterior fixation were 96% and 91%, respectively, compared with 90% and 72% for one- and two-level ACDF without anterior fixation. Similarly, Suchomel et al. [25] reported no significant difference in fusion rates and graft collapse rates between autograft and allograft protocols in either one- or two-level procedures. Although cervical plating also reduces the pseudarthrosis rate, Wang et al. [29] previously reported a high nonunion rate in three-level procedures. Thus, other strategies to increase fusion rates should be explored.
Meanwhile, although it was not statistically significant, we found the period for union was longer in allograft group (16.3 months) than in autograft group (14.1 months). If an allograft is applied as an interbody graft, new bone is derived purely from host tissues to achieve union between vertebral bodies. Therefore, it takes longer to produce enough bone formation in allograft than that in autograft. In this situation, the principal determinants of host-graft union are the stability of the construction and contact between host bone and the graft [12]. To maintain this stability, the anterior plate can share the mechanical load with graft material, and theoretically, it can also minimize subsidence by reducing the motion of the fusion segment. Suchomel et al. [25] also observed that an allograft with plate group had significantly longer time to complete union. However, there was no difference in fusion rate at the 2 years follow-up, which suggests that the increased time for union between allograft and autografts are insignificant.
Despite the anterior cervical plating, Kim et al. [4] reported that anterior cervical interbody fusion with allograft had a lower fusion rate and higher subsidence rate than that with autograft in the early period after surgery, but with similar clinical outcomes between groups. Subsidence refers to the loss of height at the operative site following surgery on the spine, caused by bone graft absorption with remodeling and graft collapse [27]. Because subsidence does not occur after union, subsidence tends to increase if union is delayed. Most studies reporting the use of allogeneic iliac crest showed high collapse rates [20]. We assumed that the shape of the interbody graft was an influencing factor of graft collapse after ACDF because a ring-shape graft has more cortical contact surface area with endplates than a tricortical-type graft, and the structure has greater capacity to resist hoop stress. It is for this reason that we used a ring-shape fibula or ulna allograft rather than tricortical-shape allograft. We believe that this shape has better resistance against graft subsidence until complete union. During the follow-up period in our study, subsidence per segment, fused segmental lordosis, and entire cervical lordosis were not different between groups (p>0.05). However, prospective comparative studies regarding graft shape are required to clarify shape effect on subsidence.
DBM does not appear to be effective and its use as a bone substitute remains controversial [13,17]. In our study, an allograft filled with DBM showed outcomes that are comparable with an autograft. Irrespective of anterior plating, one study using DBM as a graft expander in ACDF was reported >2 decades ago, comparing the outcome of allograft-DBM composite with autograft [17]. Both rates of pseudarthrosis and graft collapse were higher in the allograft-DBM group than in the autograft group without plating. After the study, few studies comparing the efficacy of DBM with autologous iliac crest bone graft in ACDF were performed. However, the use of DBM as a bone expander was supported in a recent systematic review discussing spinal fusion efficacy [30]. Although DBM and allograft has inferior biologic activity compared with autograft, additional studies are required to identify any benefit of using DBM in ACDF.
Compared with the standard autograft, an allograft filled with DBM in single- or double-level plate-supplemented ACDF surgery showed comparable clinical and radiological outcomes as well as reduced bleeding. We suggest that if a metal plate can be reinforced, an allograft is a viable alternative to autograft in ACDF surgery.
Fig. 1.
Plain lateral radiographs of a 53-year-old patient’s cervical spine. (A) Preoperative lateral view, (B) immediate postoperative lateral view, and (C, D) postoperative 1-year follow-up measurement of interspinous interval difference by flexion and extension lateral view.
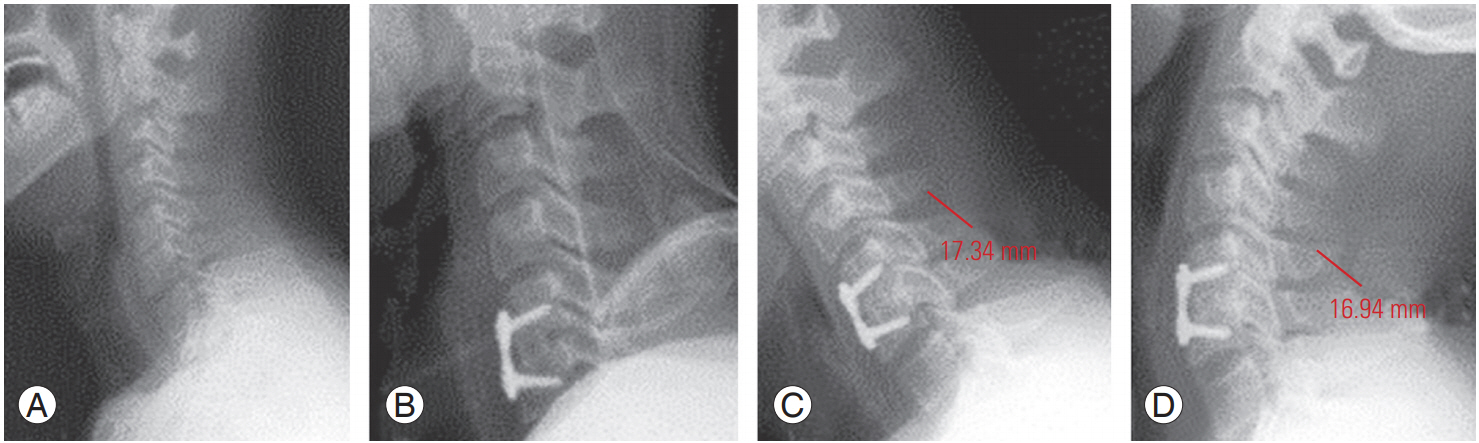
Fig. 3.
Measurement of (entire) cervical lordosis (C2 and C7) using Harrison posterior tangent method.
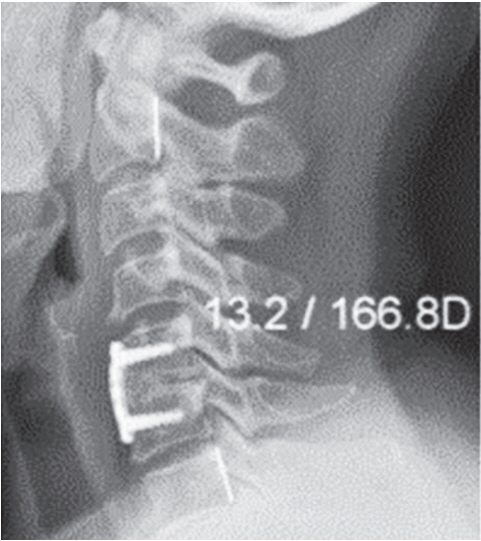
Fig. 4.
Postoperative 1-year computed tomography of C5–C6 anterior cervical discectomy and fusion. (A) Sagittal view and (B) coronal view showing continuity of bony fusion mass between C5–C6.
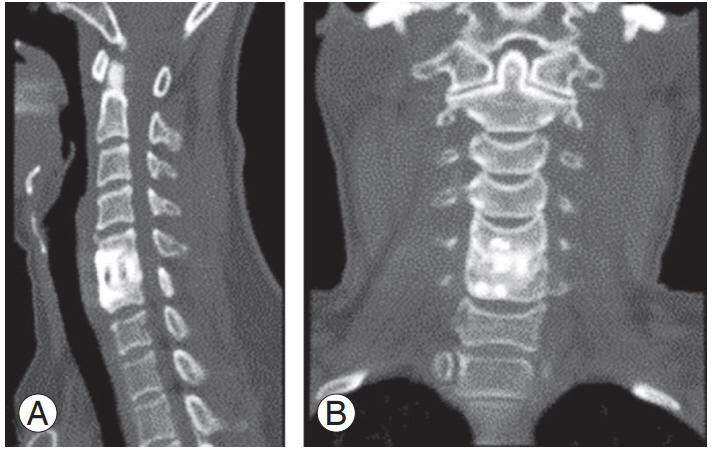
Fig. 5.
Three-year follow-up postoperative lateral radiographs of a 39-year-old patient’s cervical spine. (A) Flexion view and (B) extension view showing solid fusion of C4–C5–C6.
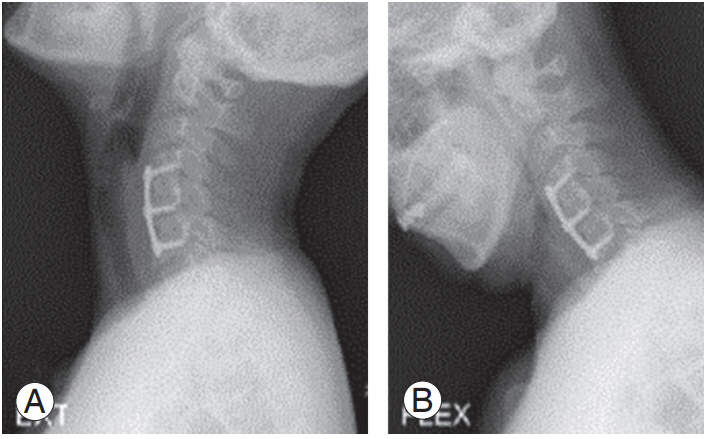
Table 1.
Patient demographics
Table 2.
Odom’s criteria
References
1. Smith GW, Robinson RA. The treatment of certain cervical-spine disorders by anterior removal of the intervertebral disc and interbody fusion. J Bone Joint Surg Am 1958 40-A:607–24.


2. Zdeblick TA, Ducker TB. The use of freeze-dried allograft bone for anterior cervical fusions. Spine (Phila Pa 1976) 1991 16:726–9.


3. Epstein NE. Iliac crest autograft versus alternative constructs for anterior cervical spine surgery: pros, cons, and costs. Surg Neurol Int 2012 3(Suppl 3): S143–56.



4. Kim SY, Park KS, Jung SS, et al. An early comparative analysis of the use of autograft versus allograft in anterior cervical discectomy and fusion. Korean J Spine 2012 9:142–6.



5. Miller LE, Block JE. Safety and effectiveness of bone allografts in anterior cervical discectomy and fusion surgery. Spine (Phila Pa 1976) 2011 36:2045–50.



6. Oh K, Lee CK, You NK, Kim SH, Cho KH. Radiologic changes of anterior cervical discectomy and fusion using allograft and plate augmentation: comparison of using fixed and variable type screw. Korean J Spine 2013 10:160–4.



7. Banwart JC, Asher MA, Hassanein RS. Iliac crest bone graft harvest donor site morbidity: a statistical evaluation. Spine (Phila Pa 1976) 1995 20:1055–60.



8. Silber JS, Anderson DG, Daffner SD, et al. Donor site morbidity after anterior iliac crest bone harvest for single-level anterior cervical discectomy and fusion. Spine (Phila Pa 1976) 2003 28:134–9.


9. Song KJ, Lee KB. A preliminary study of the use of cage and plating for single-segment fusion in degenerative cervical spine disease. J Clin Neurosci 2006 13:181–7.


10. Hilibrand AS, Carlson GD, Palumbo MA, Jones PK, Bohlman HH. Radiculopathy and myelopathy at segments adjacent to the site of a previous anterior cervical arthrodesis. J Bone Joint Surg Am 1999 81:519–28.



11. Harrison DE, Harrison DD, Cailliet R, Janik TJ, Holland B. Radiographic analysis of lumbar lordosis: centroid, Cobb, TRALL, and Harrison posterior tangent methods. Spine (Phila Pa 1976) 2001 26:E235–42.


12. Stevenson S, Emery SE, Goldberg VM. Factors affecting bone graft incorporation. Clin Orthop Relat Res 1996 (324): 66–74.

13. Campana V, Milano G, Pagano E, et al. Bone substitutes in orthopaedic surgery: from basic science to clinical practice. J Mater Sci Mater Med 2014 25:2445–61.




14. Malloy KM, Hilibrand AS. Autograft versus allograft in degenerative cervical disease. Clin Orthop Relat Res 2002 (394): 27–38.

15. Bishop RC, Moore KA, Hadley MN. Anterior cervical interbody fusion using autogeneic and allogeneic bone graft substrate: a prospective comparative analysis. J Neurosurg 1996 85:206–10.


16. Brown MD, Malinin TI, Davis PB. A roentgenographic evaluation of frozen allografts versus autografts in anterior cervical spine fusions. Clin Orthop Relat Res 1976 (119): 231–6.

17. An HS, Simpson JM, Glover JM, Stephany J. Comparison between allograft plus demineralized bone matrix versus autograft in anterior cervical fusion: a prospective multicenter study. Spine (Phila Pa 1976) 1995 20:2211–6.


18. Wright IP, Eisenstein SM. Anterior cervical discectomy and fusion without instrumentation. Spine (Phila Pa 1976) 2007 32:772–4.



19. Kurz LT, Garfin SR, Booth RE Jr. Harvesting autogenous iliac bone grafts: a review of complications and techniques. Spine (Phila Pa 1976) 1989 14:1324–31.



20. Martin GJ Jr, Haid RW Jr, MacMillan M, Rodts GE Jr, Berkman R. Anterior cervical discectomy with freeze-dried fibula allograft: overview of 317 cases and literature review. Spine (Phila Pa 1976) 1999 24:852–8.



21. Kaiser MG, Haid RW Jr, Subach BR, Barnes B, Rodts GE Jr. Anterior cervical plating enhances arthrodesis after discectomy and fusion with cortical allograft. Neurosurgery 2002 50:229–36.


22. Tuchman A, Brodke DS, Youssef JA, et al. Autograft versus allograft for cervical spinal fusion: a systematic review. Global Spine J 2017 7:59–70.



23. Samartzis D, Shen FH, Matthews DK, Yoon ST, Goldberg EJ, An HS. Comparison of allograft to autograft in multilevel anterior cervical discectomy and fusion with rigid plate fixation. Spine J 2003 3:451–9.


24. Samartzis D, Shen FH, Lyon C, Phillips M, Goldberg EJ, An HS. Does rigid instrumentation increase the fusion rate in one-level anterior cervical discectomy and fusion? Spine J 2004 4:636–43.


25. Suchomel P, Barsa P, Buchvald P, Svobodnik A, Vanickova E. Autologous versus allogenic bone grafts in instrumented anterior cervical discectomy and fusion: a prospective study with respect to bone union pattern. Eur Spine J 2004 13:510–5.



26. Zaveri GR, Ford M. Cervical spondylosis: the role of anterior instrumentation after decompression and fusion. J Spinal Disord 2001 14:10–6.



28. Ciccone WJ 2nd, Motz C, Bentley C, Tasto JP. Bioabsorbable implants in orthopaedics: new developments and clinical applications. J Am Acad Orthop Surg 2001 9:280–8.


-
METRICS

- Related articles in ASJ
-
A Review of Finite Element Modeling for Anterior Cervical Discectomy and Fusion2023 October;17(5)






