 |
 |
- Search
| Asian Spine J > Volume 17(2); 2023 > Article |
|
Abstract
Purpose
We aimed to investigate the surgical results of foramen magnum decompression (FMD) to identify the potential factors associated with syrinx reduction in Chiari malformation type I (CMI).
Overview of Literature
The predictive value of preoperative factors for syrinx reduction in patients with CMI remains debatable.
Methods
We enrolled patients who underwent microscopic FMD with outer dural layer resection for CMI. The distance from the tip of the cerebellar tonsil to the C2 vertebral endplate on sagittal magnetic resonance imaging (MRI) was defined as the tonsillar distance (TD). Patients who showed a >20% syrinx diameter reduction on the 1-year follow-up MRI were defined as the syrinx reduction group while the others were categorized in the syrinx nonreduction group. Patients with syringomyelia were categorized into the clinically improved and unimproved groups using the Chicago Chiari Outcome Scale. The imaging and clinical parameters were evaluated pre- and postoperatively.
Results
This study included 25 patients of whom 19 (76.0%) had syringomyelia. At the 1-year follow-up, the syrinx diameter had decreased in 11 patients (57.8%). The increased TD significantly differed between the syrinx reduction and nonreduction groups. At the 1-year follow-up, 12 and seven patients with syringomyelia were categorized into the clinically improved and unimproved groups, respectively. The clinically improved and unimproved groups showed significant differences in the mean age and increased TD.
Chiari malformation type I (CMI) is characterized by cerebellar tonsillar herniation through the foramen magnum, which is associated with an abnormal posterior fossa constitution [1–4]. This condition may result in syringomyelia because of subarachnoid space obstruction and cerebrospinal fluid (CSF) flow disturbance at the foramen magnum level [2,5,6]. Foramen magnum decompression (FMD) has been usually performed to relieve compression and restore normal CSF pathways at the craniocervical junction [7–11]. Some studies have reported good results ranging from obvious reduction to complete syrinx resolution after FMD [9,10,12], whereas others have described a persistent or even deteriorative syrinx after FMD [13–15].
Several preoperative factors, including sex, age, cerebellar tonsil descent degree, and preoperative syrinx size and length, have been investigated to explore their correlations with syrinx reduction following FMD [9,10,12,16]. However, the predictive value of these factors for syrinx reduction remains debatable. The alterations of hindbrain position and morphology after FMD also provided clues regarding the relationship between the hindbrain shifting and syrinx resolution after FMD. Therefore, we retrospectively investigated the surgical results of FMD by evaluating the hindbrain position alterations and attempted to identify the potential factors associated with syrinx reduction in CMI.
We enrolled consecutive patients who underwent microscopic FMD with outer dural layer resection at Hiroshima University Hospital and Hiroshima City Asa Hospital between October 2005 and April 2016 for CMI. Inclusion criteria were as follows: (1) follow-up assessments were performed for >1 year, (2) preoperative and follow-up magnetic resonance imaging (MRI) was performed 1 year postoperatively, and (3) the presence of symptoms related to CMI. Patients with syringomyelia associated with other diseases, such as spinal cord tumors and adhesive arachnoiditis, or those with spinal cord injuries were excluded from this study. This study was approved by the institutional review board of Hiroshima University (approval no., E-1960) and all patients provided informed consent.
All surgical procedures were performed in the prone position. Under general anesthesia, midline incisions extending from the inion to the upper cervical spine were performed in all patients. After a standard 4-cm-wide, 3-cm-high craniectomy of the squamous part of the occipital bone above the foramen magnum, the C1 posterior arch was resected. All patients underwent FMD combined with resection of the C1 posterior arch and outer dura mater layer, which was advocated by Isu et al. [5]. In our series, the outer dura mater layer was incised in a cross shape (Supplement 1). All patients underwent routine motor-evoked potential monitoring using transcranial electrical stimulation throughout the surgery. We used intraoperative ultrasound to observe the CSF outflow at the craniocervical junction and determine the adequacy of decompression.
CMI was diagnosed when the cerebellar tonsil descended >5 mm below the foramen magnum on midline sagittal slices on MRI [17]. We defined the distance from the tip of the cerebellar tonsil to the C2 vertebral endplate as the tonsillar distance (TD) on a T2-weighted sagittal MRI. A tonsillar herniation reduction was defined as an increased distance between the pre- and postoperative TD to calculate the changes in the hindbrain position (Fig. 1A, B). The maximal anteroposterior diameter of the syrinx and the syrinx length (defined as the number of vertebral segments spanned by the syrinx) were measured on T2-weighted axial and sagittal MRI, respectively, to evaluate the severity of the syrinx (Fig. 2A, B) [18]. These parameters were evaluated preoperatively, at 1 year postoperatively, and at a final follow-up. Patients who showed a reduction of >20% in the maximal syrinx diameter at the 1-year follow-up MRI have constituted the syrinx reduction group while the others were categorized in the syrinx nonreduction group (Fig. 3A–D) [10].
The clinical results were assessed using the Chicago Chiari Outcome Scale (CCOS) [19]. The CCOS is composed of four categories as follows: pain, nonpain symptoms, functionality, and complications. The patient is assigned a score of 1–4 in each category for a total score of 4–16, with higher numbers representing better outcomes. Each patient with syringomyelia was divided into the postoperative clinically improved group (CCOS score: 13–16) or the clinically unimproved group (CCOS score: 4–12) based on the calculated total CCOS scores [20].
The following parameters were evaluated in all patients and compared between the syrinx reduction and nonreduction groups as well as the clinically improved and unimproved groups: age upon surgery, syrinx length, preoperative coronal Cobb angle, CCOS, maximum syrinx diameter, syrinx diameter reduction, TD, increased TD, C2–C7 sagittal Cobb angle, basal and clivo-axial angles, Wiesel-Rothman interval, basion-axial interval, and atlanto-dens interval (ADI).
Values are presented as mean±standard deviation. Statistical analysis was performed using the IBM SPSS statistical software package ver. 27.0.0 (IBM Corp., Armonk, NY, USA). The Mann-Whitney U test was used to compare data between groups. Statistical significance was defined as a probability value of <0.05.
This study enrolled 25 patients (nine males and 16 females), with the mean age upon surgery and the mean duration of follow-up of 26.4±18.1 years (range, 3–66 years) and 59.0±40.0 months (range, 12–132 months), respectively. Syringomyelia was determined in 19 patients (76.0%) and scoliosis in 17 (68.0%). The mean syrinx length was 12.1±4.5, while the preoperative coronal Cobb angle was 22.7°±14.8°.
CSF leakage after surgery was found in one patient (4.0%), while none showed wound infection, neurological deficits, or vascular complications. The CSF leakage improved with conservative treatment. The mean CCOS score of our patients was 13.48±1.73, and the clinical improvement rate was 68.0% (17 patients). The mean maximum syrinx diameter and syrinx diameter reduction were 7.02±2.94 mm and 3.41±3.31 mm, respectively. The mean TD was 18.1±6.92 mm preoperatively and the TD increased by 3.73±2.88 mm at the 1-year follow-up. The mean C2–C7 sagittal Cobb angle, basal angles, clivo-axial angles, Wiesel-Rothman interval, basion-axial interval, and ADI were 19.9°±10.9°, 134.3°±7.4°, 145.1°±10.3°, 0.74±0.51 mm, 7.65±2.36 mm, and 2.04±1.49 mm, respectively (Table 1).
At the 1-year follow-up, the syrinx diameter had decreased in 11 patients (57.8%, syrinx reduction group) and remained unchanged in eight patients (42.2%, syrinx nonreduction group). At the final follow-up, syrinx reduction was observed in 13 of 19 patients (68.4%) showing upward cerebellar tonsil shifting. The mean age was 23.8±14.7 years (range, 6–53 years) and 31.8±24.2 years (range, 3–66 years) in the reduction and nonreduction groups, respectively (p=0.465). The follow-up period was 57.6±45.2 months (range, 12–132 months) and 61.5±31.1 months (range, 12–120 months) in the reduction group and nonreduction groups, respectively (p=0.457). The mean preoperative syrinx length was 10.6±5.3 and 14.1±2.4 in the reduction and nonreduction groups, respectively (p=0.135). The preoperative coronal Cobb angle was 23.7°±17.2° and 21.1°±10.6° in the reduction and nonreduction groups, respectively (p=0.865). No difference was observed in the CCOS scores between the two groups in pain symptoms (2.94±0.55 versus 2.75±0.71, p=0.431), nonpain symptoms (3.41±0.71 versus 2.62±1.06, p=0.061), and complications (3.88±0.33 versus 3.87±0.35, p=0.958). Significant differences were found in the functionality subscore (3.88±0.33 vs. 2.75±0.71, p<0.001) and the total CCOS score (14.17±1.24 versus 12.00±1.77, p=0.005). The preoperative maximum syrinx diameter was 7.08±3.74 mm and 6.98±1.84 mm in the reduction and nonreduction groups, respectively (p=0.961). The syrinx diameter reduction was 5.27±2.61 mm and 1.23±0.67 mm in the reduction and nonreduction groups, respectively (p=0.007). The mean preoperative TD was 18.74±7.15 mm and 17.21±6.91 mm in the reduction and nonreduction groups, respectively (p=0.798). The groups showed a significant difference in increased TD (5.63±2.37 mm versus 1.66±1.69, p=0.0001) but no significant differences in the C2–C7 sagittal Cobb angle (18.5°±11.5° versus 22.8°±9.7°, p=0.357), basal angle (132.5°±7.6° versus 138.1°±5.9°, p=0.092), clivo-axial angle (145.7°±11.4° versus 143.7°±8.3°, p=0.691), Wiesel-Rothman interval (0.69±0.53 mm versus 0.86±0.44 mm, p=0.424), basion-axial interval (8.18±2.09 mm versus 6.61±2.65 mm, p=0.086), and ADI (2.11±1.62 mm versus 1.87±1.12 mm, p=0.975) in preoperative radiographs (Table 2).
At the 1-year follow-up, 12 patients (63.1%) showed clinical improvement and 7 (36.9%) showed no clinical improvement with syringomyelia. The mean age was 20.9±13.2 years (range, 4–53 years) and 37.6±20.6 years (range, 3–66 years) in the improved and unimproved groups, respectively, and the patients were significantly younger in the improved group (p=0.028). The follow-up period was 52.3±40.1 months (range, 12–132 months) and 55.2±37.6 months (range, 12–84 months) in the improved and unimproved groups, respectively (p=0.736). The mean preoperative syrinx length was 11.1±5.4 and 13.7±2.2 in the improved and unimproved groups, respectively (p=0.307). The preoperative Cobb angle was 23.6°±17.2° and 21.1°±10.6° in the improved and unimproved groups, respectively (p=0.865). No difference was observed in the CCOS scores between the two groups in pain symptoms (2.92±0.61 versus 2.57±0.53, p=0.208) and complications (3.92±0.26 versus 3.71±0.49, p=0.196). Significant differences were found in nonpain symptoms (3.64±0.49 versus 2.28±0.95, p=0.002), functionality subscore (3.92±0.26 versus 2.71±0.48, p<0.001), and total CCOS score (14.47±1.01 versus 11.37±0.74, p<0.001). The preoperative maximum syrinx diameter was 7.05±3.26 mm and 6.82±1.93 mm in the improved and unimproved groups, respectively (p=0.898). The syrinx diameter reduction was longer in the improved than the unimproved group (4.42±4.04 mm versus 0.56±0.49, p=0.004). The mean preoperative TD was 18.12±7.25 mm and 17.38±7.44 mm in the improved and unimproved groups, respectively (p=0.971). The two groups showed significant differences in increased TD (5.32±2.52 mm versus 1.11±0.64 mm, p=0.0002). No significant intergroup differences were found in the C2–C7 sagittal Cobb angle (17.3°±10.9° versus 25.2°±9.4°, p=0.103), basal angle (133.5°±7.4° versus 136.2°±7.7°, p=0.477), clivo-axial angle (145.2°±11.1° versus 143.6°±11.1°, p=0.854), Wiesel-Rothman interval (0.74±0.59 mm versus 0.74±0.32 mm, p=0.853), basion-axial interval (7.05±8.38 mm versus 7.02±2.60 mm, p=0.192), and ADI (2.13±1.67 mm versus 1.85±1.11 mm, p=0.829) in preoperative radiographs (Table 3).
We investigated the potential factors associated with syrinx reduction in CMI and revealed a significant upward bulbopontine sulcus shifting after surgery in the syrinx reduction group and the clinically improved group. Additionally, we obtained favorable clinical outcomes for younger age and upward bulbopontine sulcus shifting.
FMD is widely accepted as the treatment for the syringomyelia-CMI complex. According to a recent systematic review and meta-analysis, the syrinx radiological improvement rate after FMD without a dura opening was inferior to that with a dura opening [21]. However, previous reports demonstrated that FMD without a dura opening was associated with a lower risk of complications than that with a dura opening [8,22]. We performed FMD with outer dural mater layer excision as the first choice of surgical treatment in CMI, and 68.0% of the patients showed an improvement in clinical symptoms, 57.8% showed syrinx reduction, and only one patient showed a CSF leakage. No revision surgery was performed in this study because none of the patients had serious complications or symptom deterioration postoperatively. When the symptoms get worse with a syrinx enlargement, we should consider revision surgery with duraplasty or shunt surgery. Thus, FMD with outer dural layer excision may provide optimal benefit with a low risk of postoperative complications.
Several factors, such as sex, age, length of follow-up, preoperative syrinx size, and cerebellar tonsil descent, have been reported as predictors of better surgical outcomes after FMD [9,10,12,16]. Badie et al. [16] reported that female patients showed a better response to FMD than male patients. Park et al. [9] reported that the longer the syringomyelia, the greater the syrinx improvement. Wu et al. [10] reported that patients with a more severe cerebellar tonsil descent would acquire more benefits from FMD. Our study revealed no significant differences regarding these parameters. This result may indicate that these parameters do not have a strong influence on syrinx reduction.
A few studies have attempted to evaluate preoperative craniocervical morphometric factors to predict syrinx resolution after FMD. Alperin et al. [23] suggested that physiological measures can be stronger predictors than morphological measures of posterior cranial fossa after FMD for CMI. Liu et al. [24] concluded that the morphology of the posterior cranial fossa cannot predict the response to FMD in patients with CMI. Our study revealed no significant changes in the bone morphometric parameters of the craniocervical junction, as well as the atlanto-occipital instability, such as the Wiesel-Rothman interval, basion-axial interval, and ADI. These results showed that preoperative morphological parameters did not affect syrinx reduction, as described in previous studies.
Few studies have investigated changes in the morphology of the hindbrain following FMD for CMI. Heiss et al. [25] drew a line between the basion and opisthion and selected it as the baseline and reported that pointed tonsils became round and CSF pathways significantly expanded both ventrally and dorsally at the craniocervical junction in adult patients with CMI after FMD. Xie et al. [26] selected a line crossing the clivus vertex and parallel with the C2 vertebral endplate as the baseline to evaluate the hindbrain position and showed significantly upward bulbopontine sulcus shifting. Our study selected the C2 vertebral endplate as a baseline because it was easy to measure and unchanged after FMD. As previous studies have shown, significant upward bulbopontine sulcus shifting was observed, implying that the deformed cerebellum could revert to its normal morphology with cerebellar tonsil ascent and the presence of the round tonsil. Posterior fossa decompression and upward shifting of the tip of the cerebellar tonsil may normalize the CSF flow at the foramen magnum level and facilitate syrinx resolution.
The present study had several limitations. Firstly, it is a retrospective study, which may have introduced a memory bias in the data collection. Secondly, the sample size was relatively small, which limited the study’s statistical power. Thus, future prospective large-sample studies are necessary to overcome these limitations.
Supplementary Materials
Supplementary materials can be available from https://doi.org/10.5999/aps.2022.0017. Supplement 1. Intraoperative video demonstrating surgical procedure.
Fig. 1
Tonsillar distance (TD): the distance from the tip of the cerebellar tonsil to the C2 vertebral endplate on T2-weighted sagittal magnetic resonance imaging. A tonsillar herniation reduction was defined as an increase of the distance between preoperative TD (A) and postoperative TD (B).

Fig. 2
Features of the syrinx on T2-weighted magnetic resonance imaging: maximal syrinx diameter (A) and syrinx length (B) (A) Axial; (B) sagittal. SD, maximum syrinx diameter.
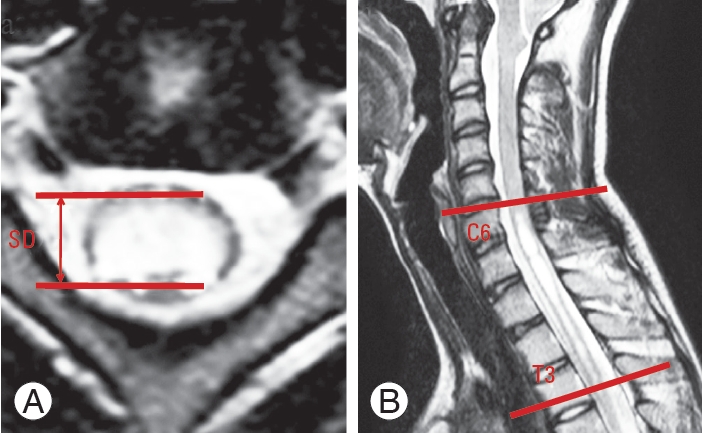
Fig. 3
Representative pre- (A, C) and postoperative (B, D) T2-weighted sagittal magnetic resonance imaging (MRI) from patients in the syrinx reduction group (A, B) and nonreduction group (C, D). Patients who showed a reduction of more than 20% in the maximal syrinx diameter at the 1-year follow-up MRI constituted the syrinx reduction group (A, B) while the others were categorized in the syrinx nonreduction group (C, D).
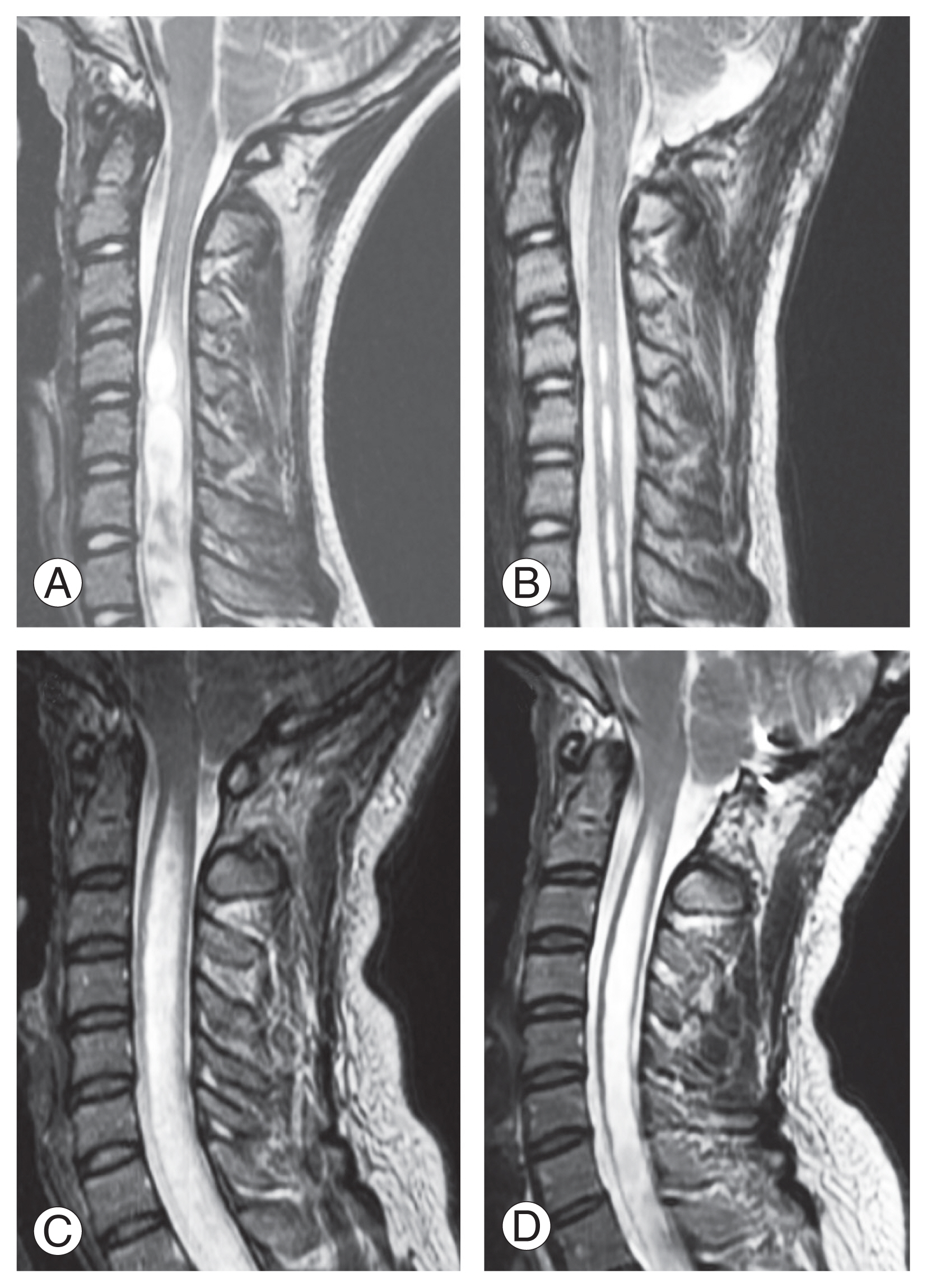
Table 1
Overall outcomes of patients who underwent foramen magnum decompression for Chiari malformation type I
Table 2
Comparison of the syrinx reduction and nonreduction groups
| Characteristic | Syrinx reduction group (n=11) | Syrinx nonreduction group (n=8) | p-value | Power |
|---|---|---|---|---|
| Age (yr) | 23.8±14.7 | 31.8±24.2 | 0.465 | 0.124 |
| Follow-up (mo) | 57.6±45.2 | 61.5±31.1 | 0.457 | 0.054 |
| Length of syrinx | 10.6±5.3 | 14.1±2.4 | 0.135 | 0.392 |
| Preoperative Cobb angle (°) | 23.7±17.2 | 21.1±10.6 | 0.865 | 0.065 |
| Chicago Chiari Outcome Scale | ||||
| Pain | 2.94±0.55 | 2.75±0.71 | 0.431 | 0.149 |
| Nonpain | 3.41±0.71 | 2.62±1.06 | 0.061 | 0.553 |
| Functionality | 3.88±0.33 | 2.75±0.71 | <0.001* | 0.993 |
| Complications | 3.88±0.33 | 3.87±0.35 | 0.958 | 0.056 |
| Total score | 14.17±1.24 | 12.00±1.77 | 0.005* | 0.801 |
| Maximum syrinx diameter (mm) | 7.08±3.74 | 6.98±1.84 | 0.961 | 0.056 |
| Reduction syrinx diameter (mm) | 5.27±2.61 | 1.23±0.67 | 0.007* | 0.986 |
| TD (mm) | 18.74±7.15 | 17.21±6.91 | 0.798 | 0.071 |
| Increase TD (mm) | 5.63±2.37 | 1.66±1.69 | <0.001* | 0.967 |
| Cervical alignment from C2 to C7 (°) | 18.5±11.5 | 22.8±9.7 | 0.357 | 0.125 |
| Basal angles (°) | 132.5±7.6 | 138.1±5.9 | 0.092 | 0.378 |
| Clivo-axial angles (°) | 145.7±11.4 | 143.7±8.3 | 0.691 | 0.068 |
| Wiesel-Rothman interval (mm) | 0.69±0.53 | 0.86±0.44 | 0.424 | 0.106 |
| Basion-axial interval (mm) | 8.18±2.09 | 6.61±2.65 | 0.086 | 0.255 |
| Atlanto-dens interval (mm) | 2.11±1.62 | 1.87±1.12 | 0.975 | 0.063 |
Table 3
Comparison of the clinical improved and unimproved groups
| Characteristic | Clinically improved group (n=12) | Clinically unimproved group (n=7) | p-value | Power |
|---|---|---|---|---|
| Age (yr) | 20.9±13.2 | 37.6±20.6 | 0.028* | 0.462 |
| Follow-up (mo) | 52.3±40.1 | 55.2±37.6 | 0.736 | 0.052 |
| Length of syrinx | 11.1±5.4 | 13.7±2.2 | 0.307 | 0.231 |
| Preoperative Cobb angle (°) | 23.6±17.2 | 21.1±10.6 | 0.865 | 0.063 |
| Chicago Chiari Outcome Scale | ||||
| Pain | 2.92±0.61 | 2.57±0.53 | 0.208 | 0.344 |
| Nonpain | 3.64±0.49 | 2.28±0.95 | 0.002* | 0.977 |
| Functionality | 3.92±0.26 | 2.71±0.48 | <0.001* | 0.999 |
| Complications | 3.92±0.26 | 3.71±0.49 | 0.196 | 0.288 |
| Total score | 14.47±1.01 | 11.37±0.74 | <0.001* | 0.999 |
| Maximum syrinx diameter (mm) | 7.05±3.26 | 6.82±1.93 | 0.898 | 0.053 |
| Reduction syrinx diameter (mm) | 4.42±4.04 | 0.56±0.49 | 0.004* | 0.735 |
| TD (mm) | 18.12±7.25 | 17.38±7.44 | 0.971 | 0.054 |
| Increase TD (mm) | 5.32±2.52 | 1.11±0.64 | <0.001* | 0.992 |
| Cervical alignment from C2 to C7 (°) | 17.3±10.9 | 25.2±9.4 | 0.103 | 0.323 |
| Basal angles (°) | 133.5±7.4 | 136.2±7.7 | 0.477 | 0.106 |
| Clivo-axial angles (°) | 145.2±11.2 | 143.6±11.1 | 0.854 | 0.058 |
| Wiesel-Rothman (mm) | 0.74±0.59 | 0.74±0.32 | 0.853 | 0.052 |
| Basion-axial interval (mm) | 7.05±8.38 | 7.02±2.60 | 0.192 | 0.054 |
| Atlanto-dens interval (mm) | 2.13±1.67 | 1.85±1.11 | 0.829 | 0.066 |
References
1. Rhoton AL Jr. Microsurgery of Arnold-Chiari malformation in adults with and without hydromyelia. J Neurosurg 1976;45:473–83.


2. Heiss JD, Snyder K, Peterson MM, et al. Pathophysiology of primary spinal syringomyelia. J Neurosurg Spine 2012;17:367–80.



3. Elster AD, Chen MY. Chiari I malformations: clinical and radiologic reappraisal. Radiology 1992;183:347–53.


4. Oldfield EH, Muraszko K, Shawker TH, Patronas NJ. Pathophysiology of syringomyelia associated with Chiari I malformation of the cerebellar tonsils: implications for diagnosis and treatment. J Neurosurg 1994;80:3–15.

5. Isu T, Sasaki H, Takamura H, Kobayashi N. Foramen magnum decompression with removal of the outer layer of the dura as treatment for syringomyelia occurring with Chiari I malformation. Neurosurgery 1993;33:845–9.


6. Schijman E, Steinbok P. International survey on the management of Chiari I malformation and syringomyelia. Childs Nerv Syst 2004;20:341–8.



7. Noudel R, Gomis P, Sotoares G, et al. Posterior fossa volume increase after surgery for Chiari malformation type I: a quantitative assessment using magnetic resonance imaging and correlations with the treatment response. J Neurosurg 2011;115:647–58.


8. Durham SR, Fjeld-Olenec K. Comparison of posterior fossa decompression with and without duraplasty for the surgical treatment of Chiari malformation type I in pediatric patients: a meta-analysis. J Neurosurg Pediatr 2008;2:42–9.


9. Park YS, Kim DS, Shim KW, Kim JH, Choi JU. Factors contributing improvement of syringomyelia and surgical outcome in type I Chiari malformation. Childs Nerv Syst 2009;25:453–9.



10. Wu T, Zhu Z, Jiang J, et al. Syrinx resolution after posterior fossa decompression in patients with scoliosis secondary to Chiari malformation type I. Eur Spine J 2012;21:1143–50.



11. Lee HS, Lee SH, Kim ES, et al. Surgical results of arachnoid-preserving posterior fossa decompression for Chiari I malformation with associated syringomyelia. J Clin Neurosci 2012;19:557–60.


12. Navarro R, Olavarria G, Seshadri R, Gonzales-Portillo G, McLone DG, Tomita T. Surgical results of posterior fossa decompression for patients with Chiari I malformation. Childs Nerv Syst 2004;20:349–56.



13. Heiss JD, Suffredini G, Smith R, et al. Pathophysiology of persistent syringomyelia after decompressive craniocervical surgery: clinical article. J Neurosurg Spine 2010;13:729–42.

14. Schuster JM, Zhang F, Norvell DC, Hermsmeyer JT. Persistent/recurrent syringomyelia after Chiari decompression-natural history and management strategies: a systematic review. Evid Based Spine Care J 2013;4:116–25.


15. Gil Z, Rao S, Constantini S. Expansion of Chiari I-associated syringomyelia after posterior-fossa decompression. Childs Nerv Syst 2000;16:555–8.



16. Badie B, Mendoza D, Batzdorf U. Posterior fossa volume and response to suboccipital decompression in patients with Chiari I malformation. Neurosurgery 1995;37:214–8.


17. Smith BW, Strahle J, Bapuraj JR, Muraszko KM, Garton HJ, Maher CO. Distribution of cerebellar tonsil position: implications for understanding Chiari malformation. J Neurosurg 2013;119:812–9.


18. Qiu Y, Zhu Z, Wang B, Yu Y, Qian B, Zhu F. Radiological presentations in relation to curve severity in scoliosis associated with syringomyelia. J Pediatr Orthop 2008;28:128–33.


19. Aliaga L, Hekman KE, Yassari R, et al. A novel scoring system for assessing Chiari malformation type I treatment outcomes. Neurosurgery 2012;70:656–64.



20. He Y, Zheng T, Wu B, Wang J. Significance of modified clivoaxial angles in the treatment of adult Chiari malformation type I. World Neurosurg 2019;130:e1004–14.


21. Perrini P, Anania Y, Cagnazzo F, Benedetto N, Morganti R, Di Carlo DT. Radiological outcome after surgical treatment of syringomyelia-Chiari I complex in adults: a systematic review and meta-analysis. Neurosurg Rev 2021;44:177–87.



22. Sakushima K, Hida K, Yabe I, Tsuboi S, Uehara R, Sasaki H. Different surgical treatment techniques used by neurosurgeons and orthopedists for syringomyelia caused by Chiari I malformation in Japan. J Neurosurg Spine 2013;18:588–92.


23. Alperin N, Loftus JR, Oliu CJ, et al. Magnetic resonance imaging measures of posterior cranial fossa morphology and cerebrospinal fluid physiology in Chiari malformation type I. Neurosurgery 2014;75:515–22.



24. Liu Z, Hao Z, Hu S, Zhao Y, Li M. Predictive value of posterior cranial fossa morphology in the decompression of Chiari malformation type I: a retrospective observational study. Medicine (Baltimore) 2019;98:e15533.








